Mitral valve repair during septal myectomy in obstructive hypertrophic cardiomyopathy
REVIEW
Mitral valve repair during septal myectomy in obstructive hypertrophic cardiomyopathy
Article Summary
- DOI: 10.24969/hvt.2023.403
- CARDIOVASCULAR DISEASES
- Published: 23/07/2023
- Received: 12/05/2023
- Revised: 19/07/2023
- Accepted: 19/07/2023
- Views: 7153
- Downloads: 4413
- Keywords: hypertrophic cardiomyopathy, septal myectomy, mitral valve repair
Address for Correspondence*: Sergei A. Budagaev, Scientific Research Institute of Cardiology and Internal Diseases, 120 Aiteke BI Street, Almaty, A05B0H6, Republic of Kazakhstan
Email: ser.bud.med@gmail.com Mobile: +77053599422 ORCID: 0000-0002-4696-4548
Sergei A. Budagaev*, Maksat A. Zhakayev, Rustem M. Tuleutayev, Michel V. Vorontsov, Artem A. Nikitenko, Zhanar N. Nurbay, Daniyar B. Kunapyanov, Marat O. Pashimov
Scientific Research Institute of Cardiology and Internal Diseases, Almaty, Republic of Kazakhstan
Abstract
Hypertrophic cardiomyopathy is a common genetic heart disease characterized predominantly by non-dilated hypertrophy of the left ventricle, in the absence of other cardiac, systemic, or metabolic diseases that can cause the observed degree of hypertrophy. The main features of left ventricular outflow tract obstruction include anatomical - basal septal hypertrophy in combination with a relatively small left ventricular cavity, leading to a change in the geometry of the outflow tract from the left ventricle, pushing the mitral valve apparatus forward, and functional - systolic anterior motion of elongated mitral valve leaflets with subsequent mitral-septal contact. Primary anatomical changes of the mitral valve make the valve more susceptible to abnormal flow vectors generated in the left ventricle cavity, creating the conditions for systolic anterior motion and mitral regurgitation. Septal myectomy, performed by experienced surgeons in specialized centers, eliminates obstruction at all levels of the left ventricular outflow tract, with a clinical success rate of 90-95% and a mortality rate of less than 1%. Some surgeons complement septal myectomy with intervention on the mitral valve, taking into account anatomical features when isolated septal myectomy may be ineffective. The choice of the most optimal concurrent intervention on the mitral valve during septal myectomy is undefined. Techniques for valve-sparing mitral valve surgery have been developed, including resection of secondary chords, edge-to-edge repair, anterior leaflet plication, and others.
This review presents the hemodynamic outcomes of concomitant interventions on the mitral valve in addition to septal myectomy in patients with obstructive hypertrophic cardiomyopathy.
Introduction
Hypertrophic cardiomyopathy (HCM) is a common (1:200 - 1:500 cases among the population) genetic heart disease characterized predominantly by non-dilated hypertrophy of the left ventricle (LV), in the absence of other cardiac, systemic, or metabolic diseases that can cause the observed degree of hypertrophy (1). The key pathophysiological determinants in HCM are obstruction of the LV outflow tract (LVOT) and diastolic dysfunction. Obstruction of the LVOT occurs in 75% of patients with HCM and manifests as shortness of breath or angina with significant functional limitations (2).
The main features of LVOT obstruction include anatomical - basal septal hypertrophy in combination with a relatively small LV cavity, leading to a change in the geometry of the outflow tract from the LV, pushing the mitral valve (MV) apparatus forward, and functional – systolic anterior motion (SAM) of elongated MV leaflets with subsequent mitral-septal contact (3). Primary anatomical changes of the MV make the valve more susceptible to abnormal flow vectors generated in the LV cavity, creating the conditions for SAM of the MV and mitral regurgitation (MR) (4). Primary anomalies of the MV apparatus include: elongated leaflets, anomalous attachment of papillary muscles directly to the anterior leaflet, and apical displacement of papillary muscles forward (5). It is important to evaluate these changes to determine the optimal strategy for invasive treatment.
Eliminating the obstruction of the LVOT through pharmacological or invasive treatment methods leads to an improvement in the clinical status (6). For patients with severe heart failure (HF) symptoms (III-IV NYHA class) that are resistant to optimal pharmacological therapy, septal reduction therapy should be considered, including septal alcohol ablation and septal myectomy (SM) (7). SM, performed by experienced surgeons in specialized centers, eliminates obstruction at all levels of the LVOT, with a clinical success rate of 90-95% and a mortality rate of less than 1% (8). Some centers achieve such results with isolated SM, while others have deemed it appropriate to include intervention on the MV (9).
Although MV replacement eliminates SAM of the MV, reducing the gradient at the level of the LVOT, it increases hospital mortality, hospitalization duration, and the risk of thromboembolic complications, which is reflected in the current AHA recommendations that call for all efforts to be made for valve-preserving surgery (10). The optimal choice of intervention for the MV in obstructive HCM not determined and often depends on the discretion of the surgeon or center.
This review aims to evaluate the results of concomitant valve-preserving surgeries during SM in HCM.
Mitral valve repair during SM in HCM
``Resect, Plicate, Release``
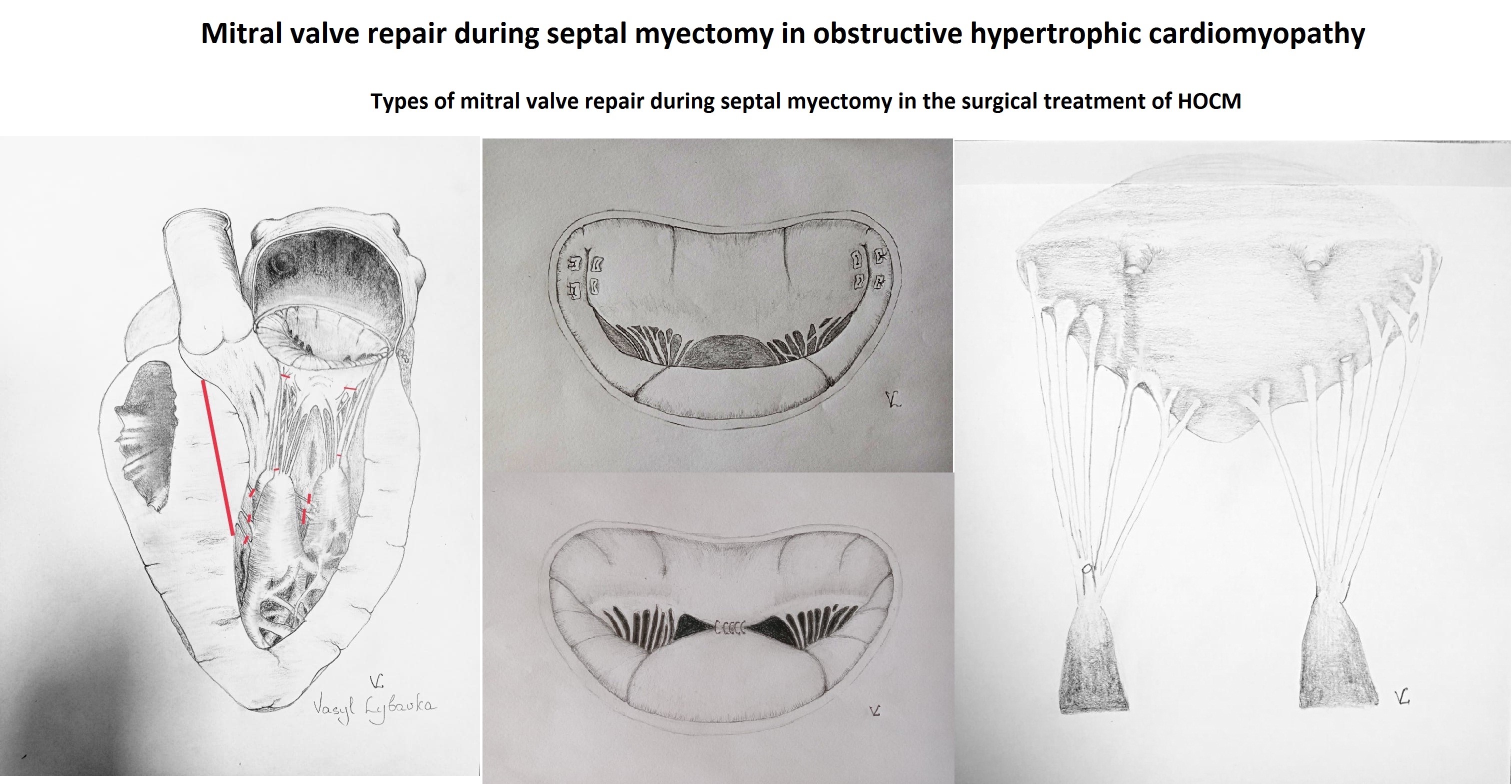
To improve the surgical outcomes of myectomy, more aggressive approaches to MV pathology have been developed, taking into account anatomical features. The "Resect, Plicate, Release" technique (Fig. 1) involves extended myectomy, plication of the anterior leaflet of the MV, and release of papillary muscles (11). The overwhelming majority of patients with obstructive HCM have elongated anterior and/or posterior leaflets of the MV compared to healthy individuals. Elongation was noted using echocardiography and magnetic resonance imaging (MRI). In obstructive HCM, the anterior leaflet is on average 34 mm compared to 24 mm in a normal heart. Elongated leaflets protrude into the LV cavity much higher than the plane of the mitral annulus, on average 26 mm higher than the annulus compared to 13 mm in a normal heart (12).
Horizontal plication of the anterior MV leaflet, developed by Dr. Swistel, makes the anterior leaflet more rigid, limiting its movement, reducing its area, shortening it, and preventing septal-mitral contact (13). Leaflet plication is achieved without disrupting the coaptation line. Furthermore, the procedure is relatively simple and can be performed through an aortotomy. In Sherrid et el. (12) retrospective analysis, which included 252 patients who underwent SM, the mean functional class of HF before surgery was 2.82. The mean resting gradient before surgery was 61 mmHg and 11 mmHg during exercise, and the interventricular septum (IVST) diameter was 24.5 mm. Plication of the anterior leaflet was performed in 66% of cases, and release of papillary muscles in 86% of cases. The operative mortality rate was 0.4%. The frequency of postoperative complications was 15%, with most being respiratory disorders in elderly patients with multiple comorbidities. The implantation of pacemaker was required in 6% of cases. Repeat surgery was required in 2 cases due to IVST defect and residual MR. A significant improvement in the functional class of HF was noted in 96% of patients, and the 10-year survival rate was 92%. Preoperative echocardiography of the anterior leaflet length of the MV provides a basis for recommending plication and determines its volume.
The authors recommend performing plication when the length of the anterior leaflet is > 30 mm or > 17 mm/m2.
Another analysis included 16 patients who underwent this procedure, with a mean gradient at the level of the LVOT of 137 mmHg and a degree of MR of 3.1, with all patients showing anterior-systolic movement (14). The postoperative gradient was 10 mmHg, and there was a significant decrease in MR to 0.2. The follow-up period was 2.4 years, during which the gradient at the level of the LVOT remained low at 6 mmHg, and the MR remained insignificant at 0.4. The subsequent follow-up was 100%, with no cases of mortality or repeat surgeries. Thus, the "Resect, Plicate, Release" method is easy to learn, relatively simple to perform, and provides excellent and durable results.
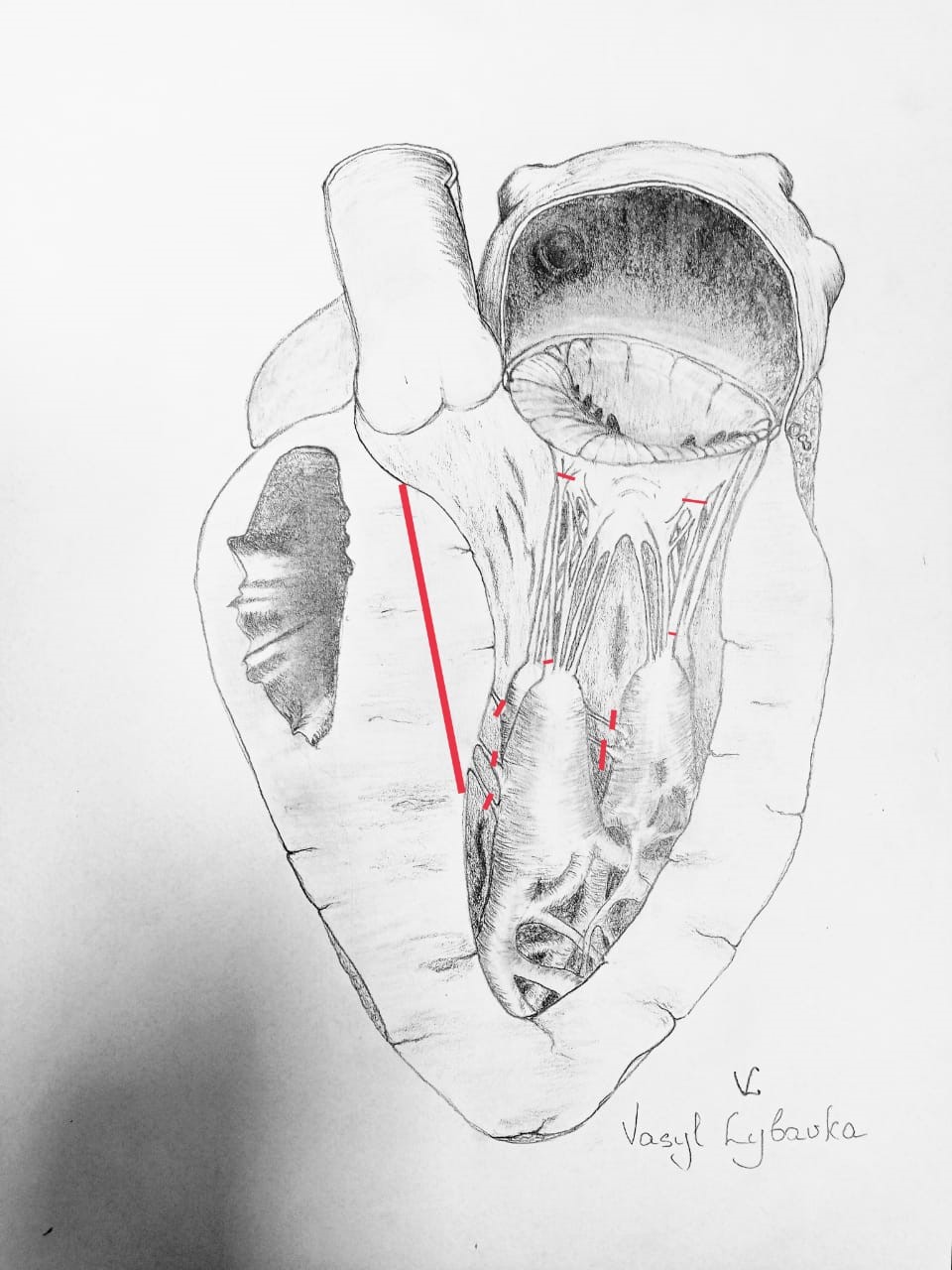
Figure 1. ``Resect, Plicate, Release``
Resection of anomalous papillary muscle
Patients with HCM have a higher frequency of papillary muscle anomalies, contributing to dynamic obstruction of the LVOT (15). In some patients, obstruction may occur without significant hypertrophy, where the only anomalies are associated with the orientation and hypermobility of the papillary muscles (16). Common findings include papillary muscle hypertrophy and anterior displacement, which result in the displacement of the MV leaflets closer to the IVST and anteriorly towards the LVOT (17). Displaced and hypertrophied papillary muscles further exacerbate the reduction in LV area, worsening the obstruction. Anomalous attachment of the papillary muscle directly to the anterior leaflet is a common anomaly of the MV in HCM and is found in approximately 13% of patients (18).
One or both heads of the papillary muscles may attach directly to the ventricular surface of the anterior leaflet of the MV. This leads to mid-ventricular obstruction (19). Increased awareness of these pathophysiological characteristics of the subvalvular apparatus of the MV and advancements in visualization methods have led to more accurate diagnosis and surgical treatment of obstructive HCM.
In a comparative analysis, Mutsuga et al. (20) evaluated the excision and reconstruction of anomalous papillary muscles in combination with myectomy. Thirty-eight patients were analyzed, and papillary muscle anomalies were present in 4 patients. The average peak gradient at the level of the LVOT was 74.3 mmHg, and the mean thickness of the IVST was 19.5 mm. In another group of 34 patients, intervention on the MV was performed in 59% of cases: 14 patients underwent MV repair and 6 patients underwent MV replacement. polytetrafluoroethylene thread was used for papillary muscle reconstruction after excision. The thickness of the IVST decreased to 4 mm in the group undergoing papillary muscle reconstructions, and the gradient at the level of the LVOT was 10.8 mmHg. All patients were in NYHA functional class I. Thus, papillary muscle reconstruction in addition to SM effectively eliminates LVOT obstruction and preserves the MV.
Anterior leaflet retention
The strategy of MV anterior leaflet retention repair (Fig. 2) developed at the German Cardiology Center in Berlin aims to prevent residual SAM syndrome (21). The results were analyzed in 25 patients with significant SAM of the MV. The average peak pressure gradient of the LVOT before the operation was 84 (32) mmHg. The mean thickness of the IVST was 23.7 mm in diastole and 26.7 mm in systole. Twenty-three patients had hemodynamically significant MR. Intraoperatively, after revision of the MV, the medial and lateral segments of the anterior leaflet closest to the fibrous triangles were fixed to the fibrous ring. The average follow-up period was 2.5 years, with a survival rate of 100% at one year after surgery and 82.6% at five years. Freedom from reoperations at five years was 83.8%. The mean peak pressure gradient across the LVOT decreased from 84.32 to 19.11 mmHg after the operation (p < 0.001), and only two patients had moderate residual or recurrent SAM of the MV during subsequent follow-up. MR and HF functional class also significantly improved during follow-up. Postoperative pressure gradient across the LVOT decreased to 19.11 mmHg. Mean systolic and diastolic IVST thickness significantly decreased during subsequent follow-up from 26.0(6.7) mm and 23.0(6.6) mm to 17.0 (4.9) mm (p=0.001) and 14.0(4.3) mm (p=0.001), respectively. At the time of discharge, MR had decreased to a negligible level in 23 patients (92%), while 4 patients (17%) continued to have mild SAM syndrome with LVOT obstruction. Recurrent SAM of the MV was observed in 2 of 17 patients during subsequent follow-up, with a mean LVOT pressure gradient of 50 mmHg and 14 mmHg, respectively. However, both patients were in NYHA functional class I.
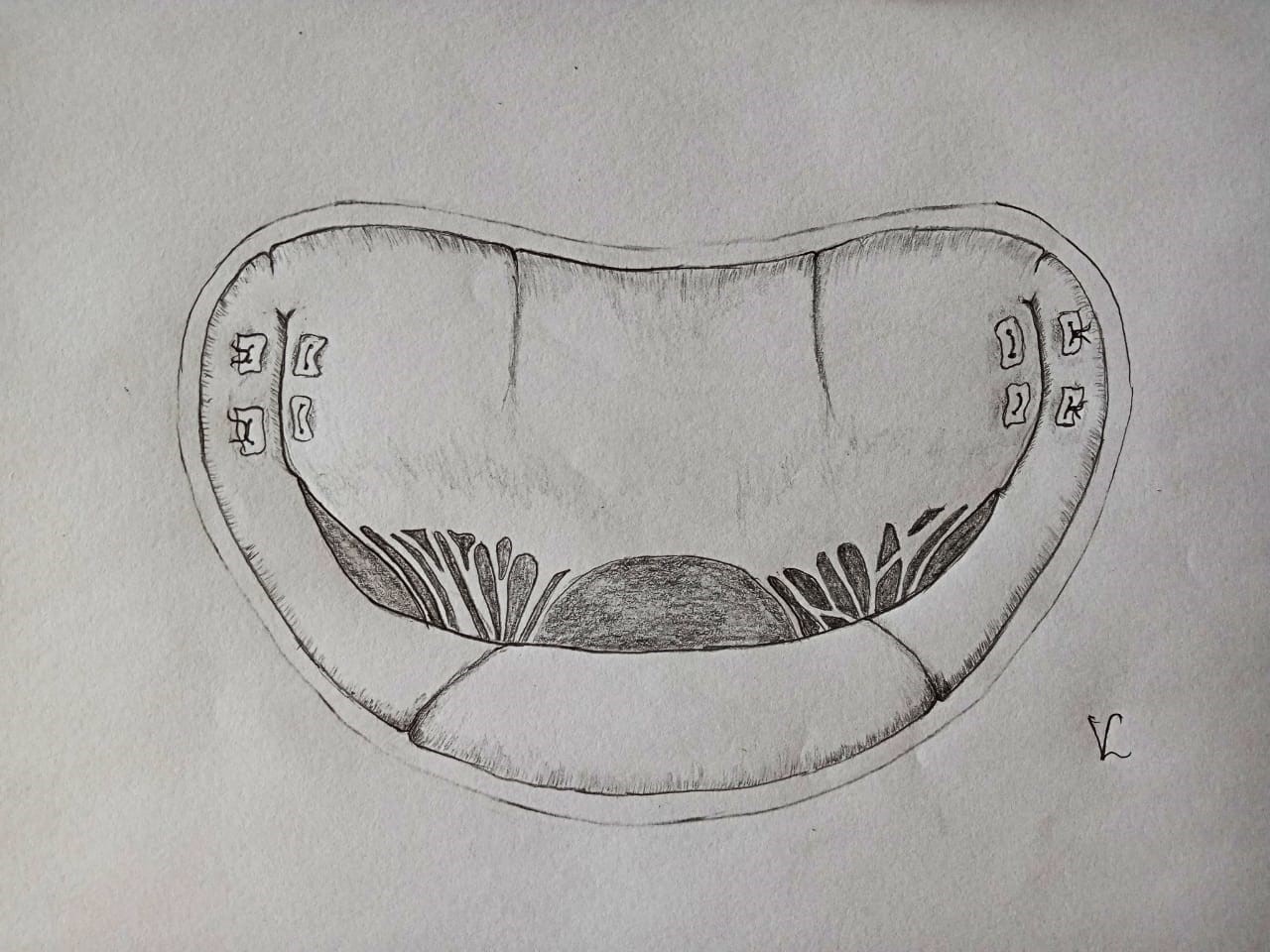
Figure 2. Anterior leaflet retention
Walter et al. presented the long-term outcomes of anterior MV leaflet retention repair in addition to SM in children (22). The intervention was performed in 12 children with a mean age of 10.8 years. The average preoperative pressure gradient at the level of the LVOT was 49 mmHg. Intraoperatively, the mean gradient at the level of the LVOT after myectomy was 5 mmHg. The mean follow-up period was 11.85 years, and the gradient at the level of the LVOT was 6.2 mmHg. There were no reported deaths, repeat myectomies, or repeat interventions on the MV.
Anterior leaflet extension
To prevent residual SAM syndrome and suboptimal outcomes of myectomy with preserved obstruction at the level of the LVOT and MR, van der Lee et al. (23) utilized a technique of anterior leaflet elongation. They demonstrated that the combined technique resulted in favorable short-term functional and hemodynamic outcomes compared to myectomy alone in these patients. A total of 29 patients underwent analysis, with anterior leaflet elongation using a patch made of processed autopericardium. The average IVST diameter before the operation was 23.4 mm. The mean gradient at the level of the LVOT was 100 mmHg. The mean follow-up period was 3.4 years, and there were no reported deaths. One patient required a repeat intervention after 3 months due to recurrent dyspnea, and a significant recurrent MR was found upon examination. Intraoperatively, suture dehiscence was identified and successfully corrected, leading to an uncomplicated clinical course. All patients were in NYHA functional class I-II after the operation. The thickness of the IVST decreased to 18 mm, and the gradient at the level of the LVOT decreased to 19 mmHg. Thus, the authors consider this method to be effective and safe in patients with obstructive HCM, and it can also be used in patients who have undergone septal ablation with recurrent LVOT obstruction.
MV repair "edge-to-edge"
The MV repair "edge-to-edge" developed more than 30 years ago has found wide application in MV surgery, including in the treatment of ischemic MR (24). The "edge-to-edge" MV repair (Fig. 3) involves fixing the central segments of the leaflets (A2, P2), thereby limiting the excursion of the anterior leaflet in the LVOT and reducing SAM of the MV (25).
A systematic review evaluating "edge-to-edge" repair in addition to SM included six studies, including five retrospective analyses and one randomized clinical trial comparing "edge-to-edge" repair and resection of secondary chords of the MV (26).
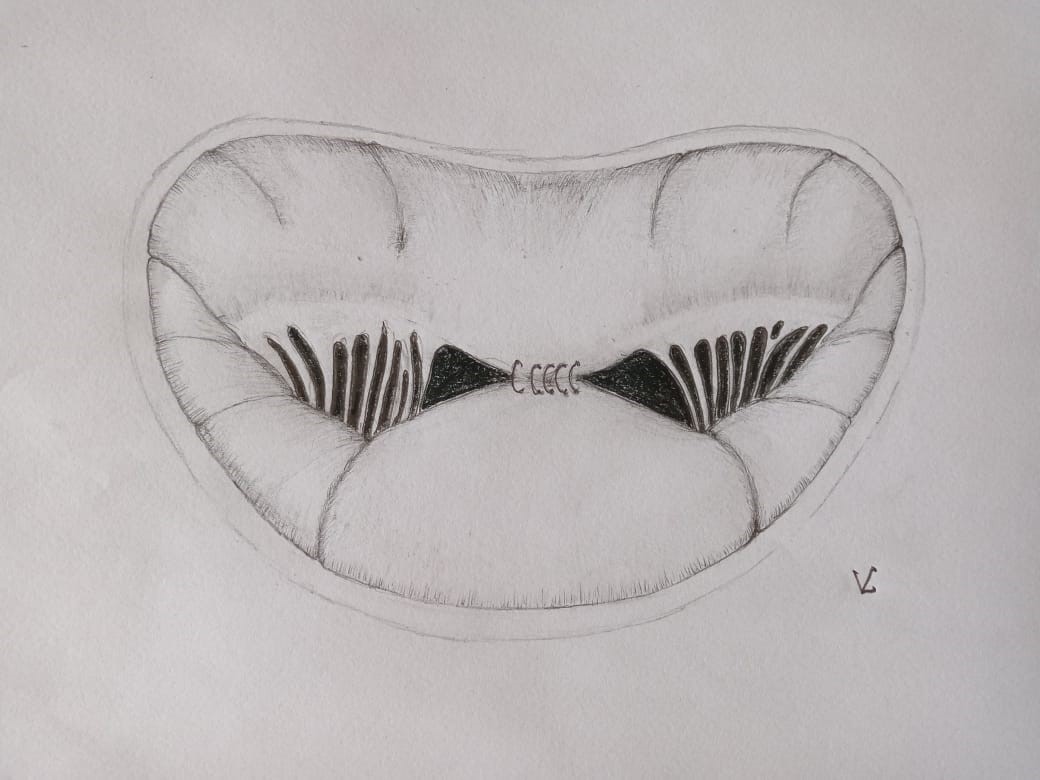
Figure 3. Mitral valve "edge-to-edge" repair
The review included 158 patients, 61% of whom had functional class III-IV HF. The mortality rate was 1% (two patients). There was a significant decrease in peak gradient at the level of the LVOT (82 (34) vs. 16 (13) mmHg, IVST diameter (24( 4) vs. 14 (4) mm), hemodynamically significant MR (84 vs. 5%), and SAM of the MV (96% vs. 0) (p<0.001 for all). The mean transmitral gradient was 4(2) mmHg. Over a follow-up period of 2.8 (2.7) years, there were 3 deaths: 2 cases of severe congestive HF and 1 case of sudden cardiac death. There were also 3 reoperations due to recurrent severe MR, and MV replacement was performed. Four patients (3%) remained in functional class III-IV HF, and 8 patients (6%) required implantation of a cardiac pacemaker. Thus, "edge-to-edge" MV repair is safe and effective in carefully selected patients requiring surgical treatment for HCM.
Sorajja proposed a percutaneous edge-to-edge MV repair technique (27). Six patients with a mean age of 83.8 years, who were deemed unsuitable for surgical intervention, underwent percutaneous edge-to-edge repair using a transcatheter system. The gradient at the level of the LVOT reduced from 91 mmHg to 12 mmHg and the MR from 3.0 to 0.8. During the subsequent 15-month follow-up, all patients showed a decrease in NYHA functional class to I-II. This initial experience suggests that percutaneous MV plication may be effective in alleviating symptoms in such patients by reducing SAM and MR.
Transaortic MV secondary chordal cutting
In patients with mild septal hypertrophy, surgeons from the Monza HCM Center proposed the use of transaortic resection of secondary mitral chordae in combination with limited SM (28). This technique (Fig. 4) is performed with the aim of reducing the gradient at the level of the LVOT, relieving symptoms of HF, and avoiding prosthetic MV replacement. Fibrotic secondary chordae plays an important role in fixing the anterior leaflet of the MV to the LVOT.
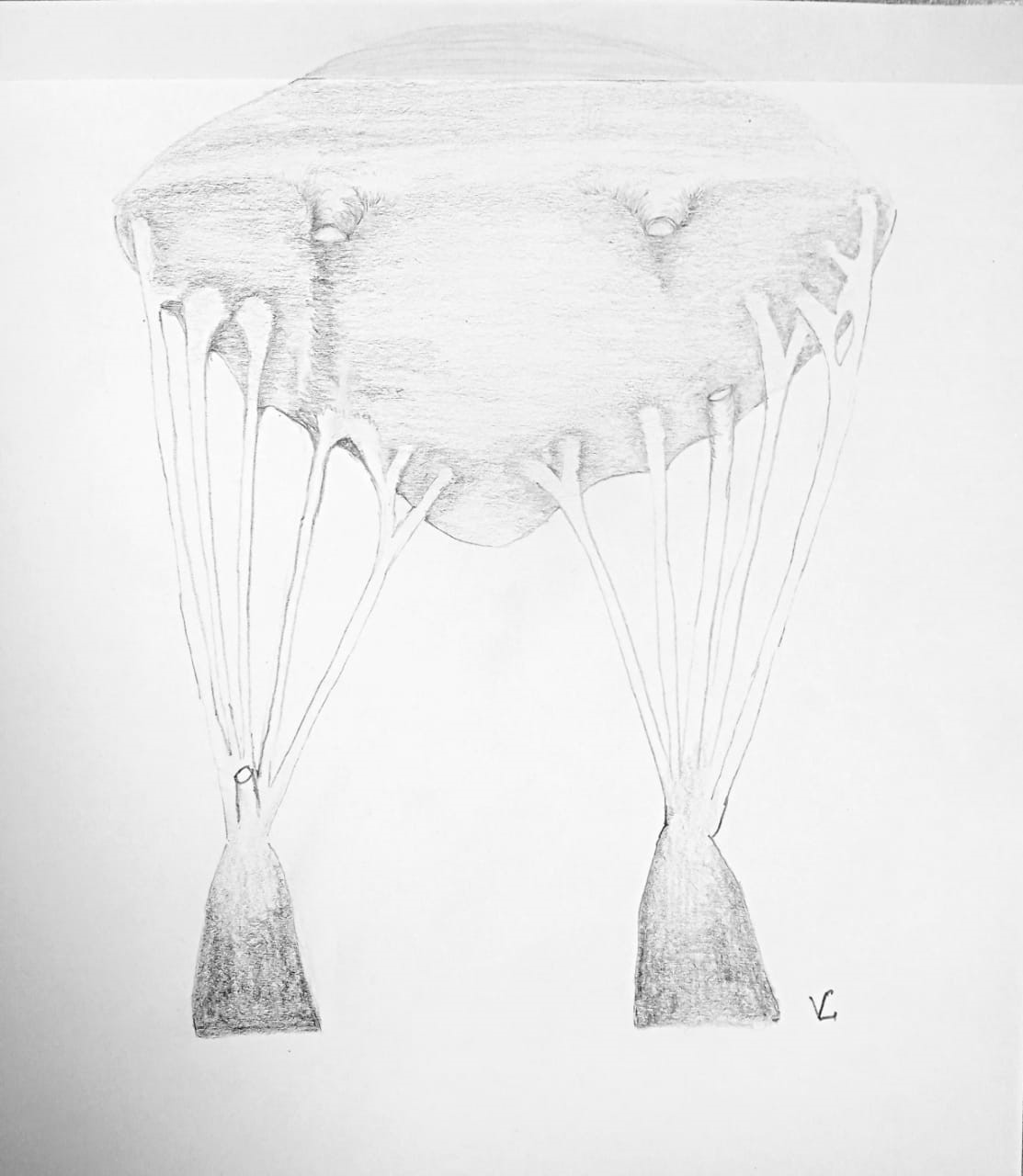
Figure 4. Transaortic mitral valve secondary chordal cutting
A single-center retrospective analysis included 39 patients with a gradient at the level of the LVOT of more than 50 mmHg and a diameter of the IVST of less than 19 mm (29). Intraoperatively, after revision of the MV apparatus due to displacement of the valve leaflet towards the left atrium, secondary chords and the tension line imposed by the restrictive chords were identified. The distal edge of the fibrous secondary chord was resected from its attachment site to the papillary muscle, while the proximal edge was resected from the ventricular surface of the anterior leaflet of the MV.
The duration of follow-up was 23.2 months, during which no patient died during hospitalization or follow-up, and no one required MV replacement. Each patient showed a significant improvement in clinical status and hemodynamics. Before surgery, 32 (82%) patients had severe symptoms of functional class III or IV, and 7 (18%) had class II. After the operation, 34 (87%) were asymptomatic in functional class I, and 5 (13%) were in class II (p<0.001). Peak gradient at the level of the LVOT before the operation was 82.5 mmHg and 9.5 mmHg after the operation (p<0.001). The diameter of the IVST was 17.1 mm before the operation and 14.2 mm after the operation (p<0.001), indicating limited SM. The technique allows for the coaptation point of the MV to be shifted to a more natural position - closer to the posterior wall of the LV and further away from the outflow tract.
A 5-year single-center experience of limited SM combined with secondary chordae resection also demonstrated excellent results, providing adequate treatment of LV outflow obstruction (30). A total of 83 patients were included, and the average diameter of the septum decreased from 24 mm to 16 mm. The average gradient at the level of the LVOT decreased from 93 (33) to 13 (11) mmHg. Hemodynamically significant MR was observed in one patient in the postoperative period, out of 32 (39%) before the operation.
Discussion
SM, developed in 1968, has undergone several changes and allows elimination of obstruction at any level of the LVOT with optimal hemodynamic results (31). The combination of basal hypertrophy and primary anomalies of the MV overall promotes SAM of the MV, which ultimately determines the obstruction of the LVOT anomalous coaptation of the MV leaflets, and MR (32).
Extended SM is the method of choice for obstructive HCM and is usually sufficient to eliminate obstruction at the level of the LVOT, SAM of the MV, and MR (33). The adult cardiac surgery database of the Society of Thoracic Surgeons indicates that 34% of cases require concomitant MV intervention during myectomy in the United States, of which 60% are valve-preserving procedures and 40% are MV replacement (34). In this database, 500 concomitant MV interventions were performed, with an operative mortality of 2%, 5% requiring pacemaker implantation, and 0.6% developing ventricular septal defects. In 67 patients with HCM and congenital MV disease at the Cleveland Clinic, the operative mortality was 3%, freedom from moderate or severe recurrent MR at 1 year was 90%, and 1-year and 5-year survival rates were 91% and 81%, respectively (35).
There is a risk of residual SAM of the MV, primarily associated with MV anomalies that displace the MV apparatus towards the left atrium, causing obstruction and MR (36). This is particularly important if there is mild septal hypertrophy, where isolated SM may be ineffective and further intervention is associated with a high risk of iatrogenic septal defects. Primary MV anomalies such as leaflet elongation and/or fibrosis, secondary chordal restriction, anomalous papillary muscle attachment, and others can be effectively and safely corrected during SM (37). Patel et al. (38) demonstrated that a concurrent MV procedure is indicated in a significant proportion of patients with a basal septal thickness of less than 18 mm. Hodges et al. (39) showed that as the degree of hypertrophy decreases, MV intervention becomes an increasingly important component of the surgical management of HCM. Furthermore, about 10% of patients with HCM have structural MV abnormalities causing severe MR - degenerative and restrictive anomalies (40).
To correct anomalies of the MV, the simplest available technique that requires minimal time should be used to avoid complications in an already complex operation. These surgical interventions can be performed via transaortic access, thereby reducing the risks and time associated with additional transatrial access. However, surgical treatment of associated MV disease remains a subject of debate. The choice of the most optimal method for correcting MR in HCM is not defined and is mainly determined by the preferences of the surgeon or center.
Visualization methods such as transesophageal echocardiography and cardiac MRI allow more accurate diagnosis of various pathologies of the MV apparatus at all levels and can be useful in determining the most effective surgical treatment strategy.
Study limitations
The methods for correcting MR were performed on a relatively small number of patients and with a short-to-medium-term follow-up period. The studies are often retrospective in nature and there is often no comparison group – patients with isolated SM.
Conclusion
Additional methods for correcting MR are effective and safe for well-selected patients with HCM and primary MV anomalies. They allow elimination of LV outflow obstruction, even in patients with mild septal hypertrophy, thereby reducing the risk of septal defects, LV rupture, and other complications. These methods also allow the elimination of SAM of the MV, MR, and preservation of the valve, avoiding complications associated with MV replacement. Further research on associated interventions on the MV is necessary to determine the optimal surgical approach for HCM patients.
Peer-review: External and Internal
Conflict of interest: None to declare
Authorship: S.A.B., R.M.T., M.V.V., A.A.N., Z.N.N., D.B.K., M.A.Z., M.O.P. equally contributed to the study and preparation of manuscript
Acknowledgement and funding: This study was conducted as part of the scientific and technical program BR 11065383 «Development of innovative and highly efficient technologies to reduce the risk of premature mortality from diseases of the circulatory system, chronic respiratory diseases and diabetes”.
References
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Copyright

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
AUTHOR'S CORNER

