A rare case of free floating thrombus in the aorta and left ventricle – a case report
CASE REPORT
A rare case of free floating thrombus in the aorta and left ventricle – a case report
Article Summary
- DOI: 10.24969/hvt.2023.412
- Page(s): 387-392
- Cardiac Surgery
- Published: 22/08/2023
- Received: 09/04/2023
- Revised: 16/08/2023
- Accepted: 17/08/2023
- Views: 5623
- Downloads: 4460
- Keywords: : aorta thrombus, left ventricular thrombus, surgical thrombectomy
Address for Correspondence*: Nihaz Yusuf Nazer, Department of Cardiothoracic and Vascular Surgery
Sri Jayadeva Institute of Cardiovascular Sciences and Research, Bangalore, Karnataka, 560069, India
Mobile: +9188800555 E-mail: nihaznazer@gmail.com
Nihaz Yusuf Nazer1*, Shilpa Suresh1, Jyothirmayi Bommali2, Ashita Barthur2, Ravi Kishore3, Rajesh K. Rao1
Department of Cardiothoracic and Vascular Surgery1, Department of Radiology2 and Department of Cardiology3, Sri Jayadeva Institute of Cardiovascular Sciences and Research, Sri Jayadeva institute of cardiovascular sciences and research, Bangalore, Karnataka, India
Abstract
Objective: Aortic thrombi, though rare, represents a significant medical entity requiring precise diagnosis and management. We describe an intriguing case of cardiac syncope resulting from an free-floating thrombus in the aorta.
Case presentation: A 27-year old male smoker presented with recurrent episodes of syncope. Diagnostic investigations (echocardiography, computed tomography, magnetic resonance imaging) unveiled the presence of thrombi within the ascending aorta and the left ventricle. The patient underwent successful surgical removal of free-floating thrombus in aorta and left ventricle. The proper anticoagulation was initiated before and continued after surgery along with antiaggregant therapy to prevent embolization and recurrence.
Conclusion: Given the lack of extensive data, the management of aortic thrombosis remains variable, encompassing strategies ranging from anticoagulant pharmacotherapy to surgical intervention, or a combination of both. The absence of established guidelines further underscores the intricacies in the therapeutic approach to this condition. We delineate the clinical trajectory of the patient who underwent successful surgical excision of the thrombus considering the risk of cerebral and distant embolic phenomenon, which may be life threatening,
Key words: aorta thrombus, left ventricular thrombus, surgical thrombectomy
Introduction
Free-floating thrombus (FFT) in the aorta is a rare entity. It may occur in the absence of any pre-existing aortic disease or coagulopathy but is usually seen in patients with steroid abuse, drug overdose, blood dyscrasias, trauma and vasculitis (1). Idiopathic FFT may remain silent and go unnoticed (2). However, they can be life-threatening due to cerebral, systemic or peripheral embolism. The cause is frequently unknown and elusive, and computed tomography (CT) angiography is the diagnostic modality of choice. There are no established guidelines for the management of FFT.
Here, we describe an intriguing case of cardiac syncope resulting from an FFT in the aorta.
Case report
A 27-year-old male presented to our emergency department with history of recurrent syncope for the past 2 months. He complained of three syncopal episodes in the past two weeks. Except for smoking, there were no other risk factors including other comorbidities and a negative history for coronavirus disease and drug abuse.
Patient’s sensorium and vitals were within normal at presentation. Peripheral arterial assessment revealed diminished pulses in the lower extremities. Rest of systemic examination was normal.
Electrocardiogram indicated prior inferior wall myocardial infarction based on q waves in the inferior leads. Initial blood investigations including troponin T and fasting lipid profile were unremarkable. Thrombophilia screening was done after results of magnetic resonance imaging (MRI), of which homocysteine level was 17 micromol/L (normal <15 micromol/L). Subsequently, hematology consultation was done and it was advised that the elevated value was not clinically important.
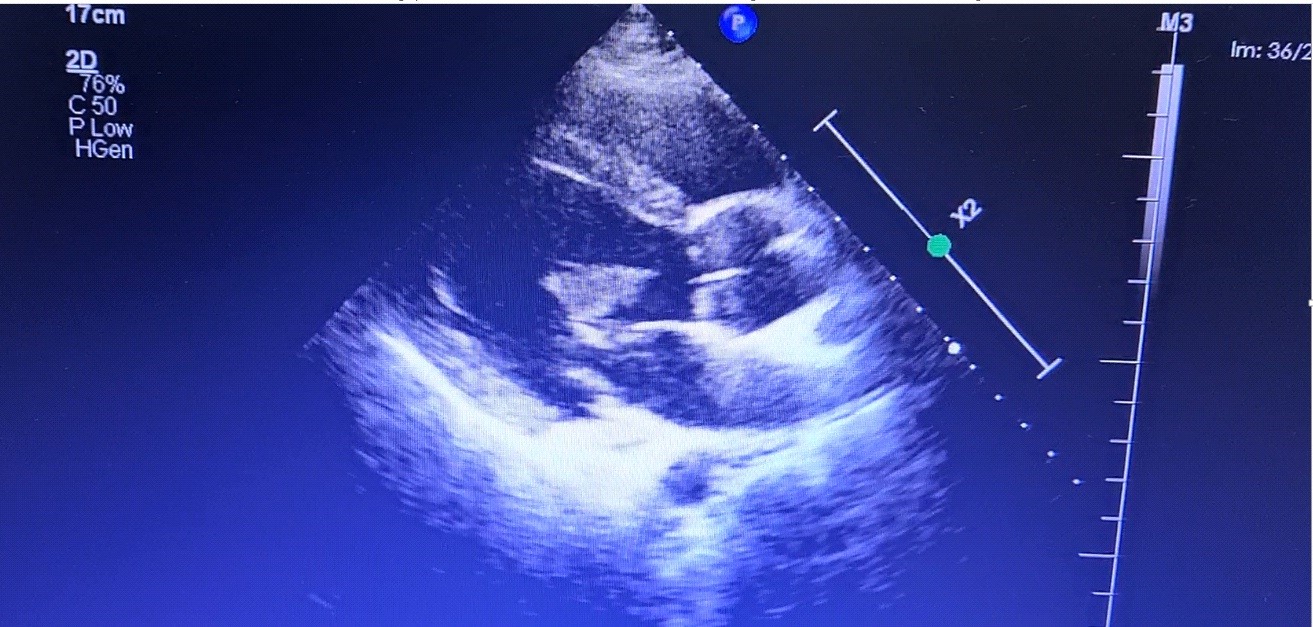
Figure 1. Transthoracic echocardiography showing the mass in left ventricle (white arrow) and a suspicious echogenicity in the ascending aorta (red arrow)
Transthoracic 2-dimensional echocardiogram (TTE) demonstrated regional wall motion abnormality with a left ventricular (LV) ejection fraction of 55%.There was an aneurysm in the inferobasal portion of the LV from which a pedunculated mass was noted to arise (Fig. 1). In addition, a suspicious mass was also noted in the aortic root.
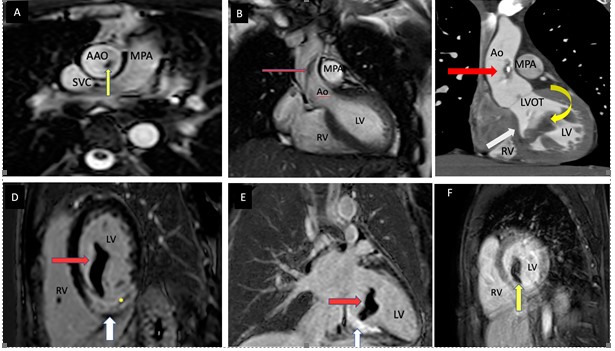
Figure 2. Magnetic resonance imaging: (A) – BTFE axial sequence showing floating thrombus (Block yellow arrow) in AAo. (B) still frame of a BTFE coronal cine sequence showing floating thrombus (Block arrow) in AAo. (C) Oblique coronal view partially showing floating thrombus in AAo with speck of calcification (Block red arrow) and LV thrombus (curved yellow arrow) attached along interventricular septum with RCA territory infract (Block white arrow). (D) PCIR short- axis showing RCA territory infarct (Block white arrow), with microvascular obstruction (yellow asterisk) and LV thrombus (red arrow). (E) PSIR 2-chamber view showing RCA territory infarct (Block white arrow) with a floating pedunculated thrombus (Block red arrow) attached to the infarcted segment. Fig (F). Long TI image in short- axis (TI 600ms) showing the hypointense floating thrombus.
AAo - ascending aorta, BTFE - balanced turbo field echo () LV- left ventricle, PSIR -phase-sensitive inversion recovery RCA- right coronary artery
To further delineate the pathology, a cardiac MRI was performed. In the ascending aorta, a 34 x 6.5 mm free floating thrombus attached to the right aortic wall at sinotubular (ST) junction above right coronary cusp was noted (Fig. 2A-C).
A large pedunculated freely mobile mass (11x17x35mm) was found to be attached to the mid inferoseptal segment of the LV and extending into the LV outflow tract (LVOT) (Fig. 2C-F). There was also an infarct in the right coronary artery (RCA) territory (Fig. 2D).
Given the risk of embolism associated with invasive coronary angiography, a CT coronary angiogram (CT CAG) was performed to assess the coronary artery anatomy. CT CAG showed diffuse luminal irregularity in proximal RCA causing moderate luminal narrowing (40-50%). CT of brain was done to rule out infarctions and was normal.
Peripheral Doppler ultrasonography was performed, which showed triphasic flows up to bilateral popliteal arteries, monophasic flows in anterior tibial arteries and absent of color uptake in posterior tibial arteries and dorsal pedal arteries. Vascular consultation was done and medical management for peripheral vascular occlusive disease and life- style modification with smoking cessation was advised.
Once the diagnosis of thrombus was established with the MRI findings, the patient was initiated on unfractionated heparin infusion and dose was titrated to maintain a partial thromboplastin time (PTT) twice the normal value.
Due to the risk of embolic complications, a multidisciplinary decision favored an early surgical intervention as opposed to medical therapy or a delayed surgery. The patient was taken up for surgical extraction of the LV and aortic thrombi after obtaining informed high- risk written consent.
Following a standard median sternotomy, the pericardium was opened, taking care not to manipulate the aorta significantly. Intraoperatively, an epicardial echocardiography was performed to guide the approach and to aid the cannulation. Aorta was opened under fibrillatory arrest and then clamped proximal to innominate artery, avoiding the clot. A pedunculated mass was noted 2 cm above the right coronary cusp and non-coronary cusp (Fig. 3), with an otherwise normal appearing aorta, which was removed. Through the same incision, the LV was visualized, which revealed a 6 x 4 x 4 cm clot attached by a pedicle to the anterior mitral leaflet chordae (Fig. 4). The LV clot was carefully extracted in-toto. The complete extraction was confirmed by videoscopy. The aortic valve showed no signs of any pathology and it was preserved.
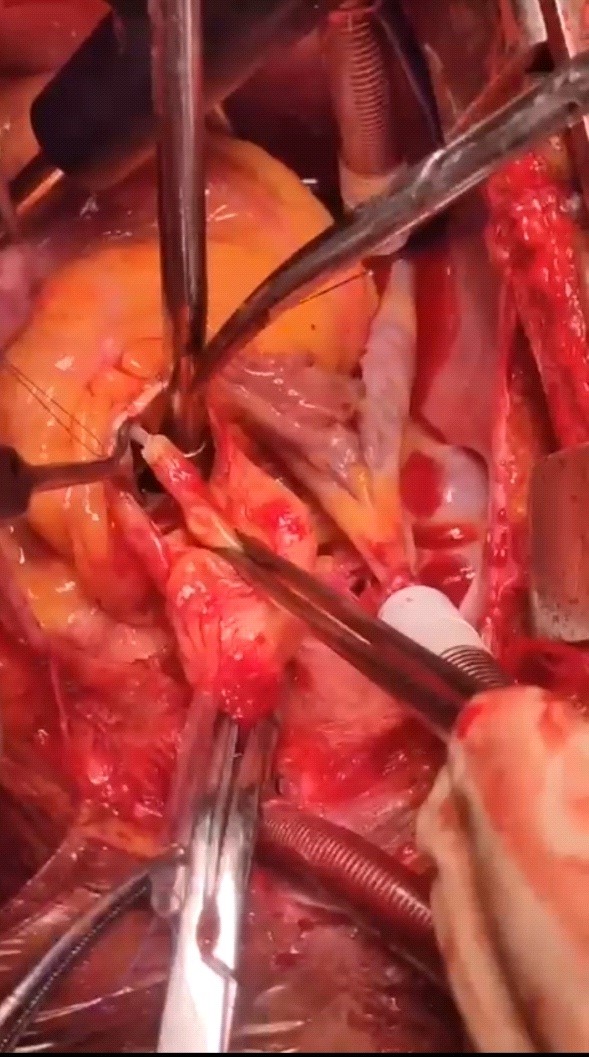
Figure 3. Thrombus in the ascending aorta (white arrow) with stalk attached to anterior part of aorta near the right coronary sinus (as seen from surgeon’s perspective)
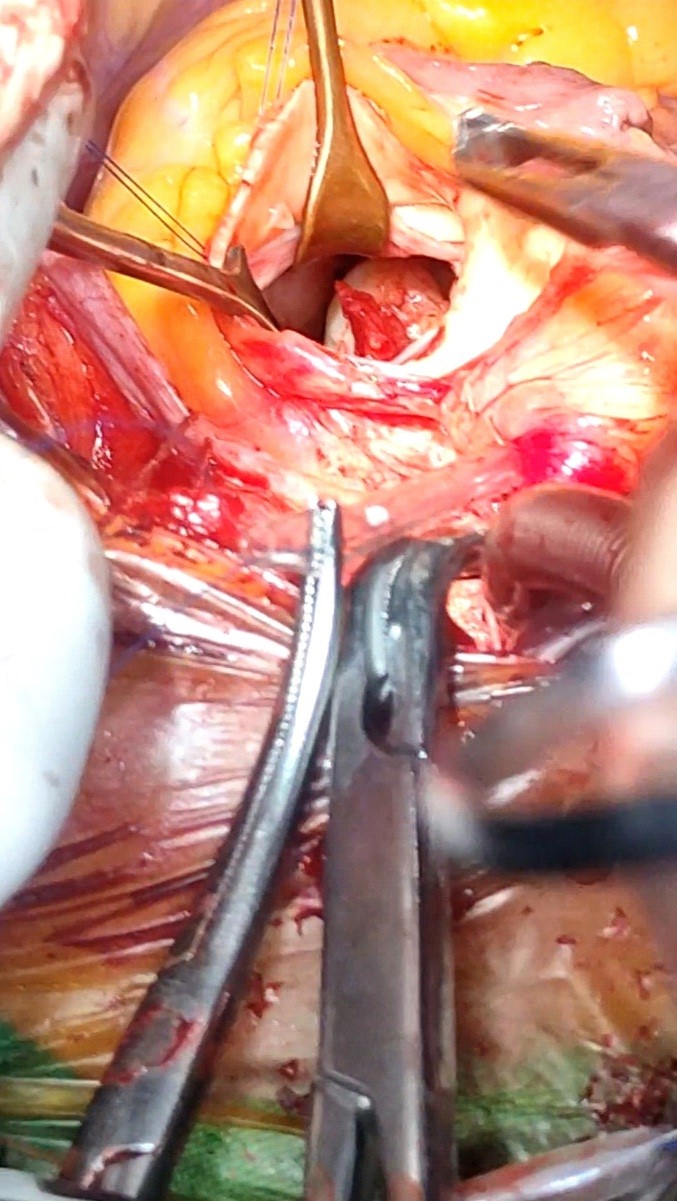
Figure 4. Thrombus in the left ventricle (white arrow) attached by a pedicle near the anterior mitral leaflet chordae as visualized though the aorta from the surgeon’s side.
Post-operative period was uneventful. Patient was started on unfractionated heparin at 5000U intravenously thrice daily along with warfarin; and heparin was stopped when INR reached therapeutic range between 2 to 3.
The patient was discharged with stable hemodynamics on warfarin and aspirin 75 mg once daily. Histopathology was suggestive of an organized thrombus. A follow-up echocardiography immediate post operation and at 3 months follow up showed no evidence of a residual mass.
Discussion
We presented a case of surgical removal of LV thrombus and free-floating thrombus in aorta, established by diagnostic imaging as echocardiography, MRI and CT. The proper anticoagulation was initiated before and continued after surgery along with antiaggregant therapy prevent embolization and recurrence.
The available evidence and literature on free-floating thrombus in ascending aorta are limited and dispersed. In a study done by Machleder et al. (3), the incidence of FFT in the thoracic aorta was found to be 0.45% in a large study of 10,671 autopsies. Karalis and colleagues (4) reported a 73% incidence of embolic events among patients with pedunculated and highly mobile aortic thrombi.
The management depends mainly on the thrombus's location and inherent morphology (3). Intramural thrombi have less risk of embolization than pedunculated or cylindrical thrombi (4). Management options include anticoagulation, thrombolysis or surgical extractio .
In those thrombi which carry a relatively higher risk of embolization, surgical thrombectomy is the preferred therapy. Choukroun et al. (5) recommend that FFT in ascending aorta and the aortic arch is an indication for surgery. The location of the thrombus also affects the surgical technique- the cannulation site, need for deep hypothermic circulators arrest (DHCA) and placement of the cross-clamp (2).
In studies comparing surgery versus medical therapy with anticoagulation, some authors have advised that conservative management can be considered in asymptomatic patients, with repeat scans and hospitalization till significant reduction in thrombus size is documented whereas others have found that while using anticoagulation as the primary therapy, recurrence rates and complications were higher (6).
Even with surgery, post-operative optimization of the anticoagulation therapy plays a vital role in preventing recurrences. There is no standardized evidence regarding the optimal PT-INR in such patients. However, a study by Oki et al. (2), suggested that an INR of 2 was sufficient to prevent recurrences.
Rarer studies exist with regard to surgical management of thrombus in the left ventricle but some authors have suggested that that early thrombectomy should be considered when a mobile, pedunculated left ventricular thrombus is demonstrated by two-dimensional echocardiogram (7).
While there exists an inherent risk of embolism with the conservative line of management, the risk of an aortic thrombectomy is low, even when hypothermic circulatory arrest is required (8). Some surgeons have even advocated that replacement of the weak portion of aorta could represent a valid and effective treatment for reducing recurrence rate of thrombi (9).
Newer modalities such as transesophageal echocardiography guided thrombi exclusion using endovascular stented grafts are being studied but the risk of thrombi fragmentation and thrombogenicity of stent are still unidentified (10).
The Task Force for the Diagnosis and Treatment of Aortic Diseases of the European Society of Cardiology (11) has recommended that with regard to thrombus in the aorta, the treatment consensus should be based on experts’ opinion (level of evidence C) and every case becomes peculiar and must be discussed in a Heart Team.
Conclusion
FFT in the ascending aorta is a rare entity but can result in life threatening complications. The choice between anticoagulation and surgical management depends on a multitude of factors and there is no gold standard treatment.
A multidisciplinary heart team discussion is warranted to tailor the appropriate treatment to each case. While there is no clear guidelines to adhere by, we have learned the following: a through pre-operative evaluation is necessary to try to determine the cause of thrombus and appropriate use of imaging modalities (TTE, CT, MRI) to precisely identify the location of the thrombus. An intraoperative epicardial or transthoracic echocardiography helps to guide the surgeon.
As demonstrated in our case, early surgical intervention is a feasible and safe option. While there is no consensus on the optimal target range for anticoagulation, anticoagulation is essential in preventing recurrences.
Take home message: We delineate the clinical trajectory of the patient with syncope, which was caused by free-floating thrombi in aorta and LV, established by imaging modalities as TTE, CT and MRI, who underwent successful surgical excision of LV and aortic free-floating thrombi considering the risk of cerebral and distant embolic phenomenon, which may be life- threatening. The proper anticoagulation and antiaggregant therapy is necessary to prevent embolization and recurrence.
Ethics: Informed consent was obtained from patients before all procedures
Peer-review: External and Internal
Conflict of interest: None to declare
Authorship: N.Y.N., S.S., J.B., A.B., R.K., and R.K. R. contributed equally to the Case management and preparation of manuscript
Acknowledgement and funding: None to declare
References
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Copyright

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
AUTHOR'S CORNER

