Right coronary artery thrombosis within drug eluting stents more than 10 years after deployment: Two cases of very, very late stent thrombosis
CASE REPORT
Right coronary artery thrombosis within drug eluting stents more than 10 years after deployment: Two cases of very, very late stent thrombosis
Article Summary
- DOI: 10.24969/hvt.2017.20
- Page(s): 58-60
- CARDIOVASCULAR DISEASES
- Published: 11/09/2017
- Received: 04/09/2017
- Revised: 09/09/2017
- Accepted: 11/09/2017
- Views: 11324
- Downloads: 9370
- Keywords: percutaneous coronary interventions, stent, thrombosis
Address for Correspondence: Sam Lovibond,
MonashHeart, Melbourne, Victoria, Australia, E-mail: slovibond@gmail.com
Authors
MonashHeart, Melbourne, Victoria, Australia
MonashHeart & Melbourne Heart Care, Melbourne, Victoria, Australia
Abstract
We present two rare cases of very late stent thrombosis occurring beyond ten years post initial drug eluting stent deployment.
Stent thrombosis (ST) is a known severe complication following percutaneous coronary intervention (PCI). We present two rare cases of very late stent thrombosis (VLST) occurring beyond ten years post initial drug eluting stent (DES) deployment, which has not previously been documented. Additionally, one patient experienced recurrent episodes of VLST within the same vessel. The cases show features of multiple contributory risk factors for VLST, which help highlight its pathogenesis and mechanisms for prevention.
Case 1
A 50-year old man suffered a non-ST-elevation myocardial infarction (NSTEMI) in 2005. Coronary angiography (CA) revealed a thrombotic right coronary artery (RCA) occlusion. Three TAXUS (Boston Scientific) stents were deployed. Aspirin, clopidogrel and secondary prevention medications were commenced.
In 2007 he was re-admitted to hospital with NSTEMI following clopidogrel cessation. CA revealed ST within his RCA stents with significant positive remodeling. Thrombus aspiration only was performed and clopidogrel was restarted.
In September 2015, he represented with an inferior ST-elevation myocardial infarction (STEMI) following self-ceasing clopidogrel and aspirin seven days prior. CA demonstrated ST (Fig. 1). Following clot aspiration, balloon angioplasty only was undertaken. Intravascular coronary ultrasonography (IVUS) examination of the stented portion of the vessel revealed significant positive remodeling of the artery (Fig. 2, blue arrow for online and cover version, grey in print version) away from the stent (Fig. 2, red arrow for online and cover version, dark grey in print version) resulting in substantial malapposition. Bypass grafting to the distal RCA was subsequently undertaken, with mid-vessel ligation to prevent competitive flow.
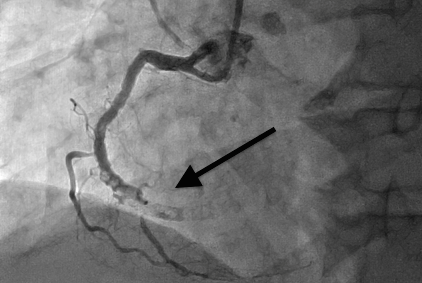
Figure 1. Left anterior oblique (LAO) view of the RCA displaying severe positive remodeling around the proximal stent and distal stent occlusion (arrow).
RCA- right coronary artery
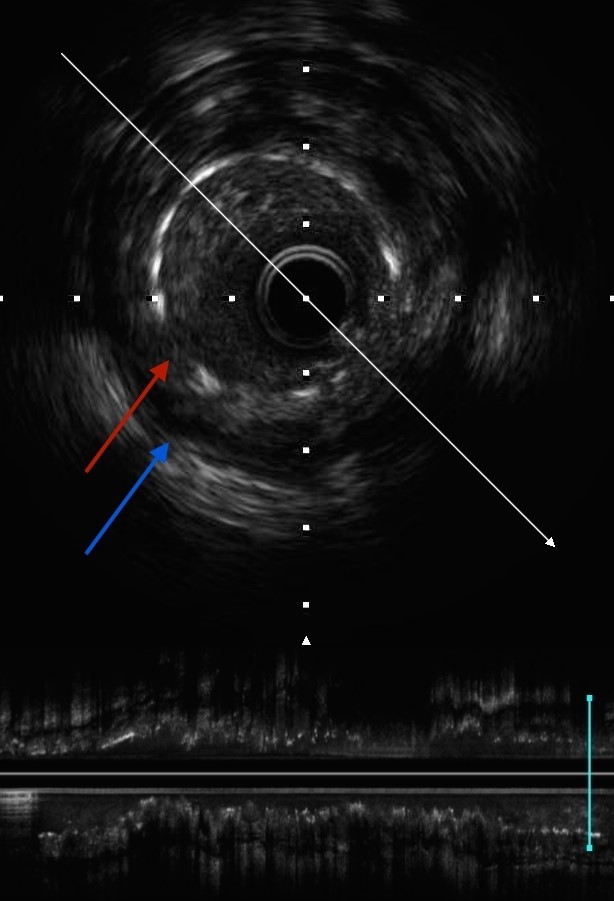
Figure 2. IVUS examination of the stented portion of the vessel (from Image 1) revealing significant positive remodeling of the artery (blue arrow) away from the stent (red arrow) resulting in malapposition.
IVUS – intravascular ultrasound
Case 2
A 72-year old man suffered an inferior STEMI in 2004. CA revealed a severe mid RCA stenosis and a TAXUS DES was deployed. Aspirin, clopidogrel and secondary prevention medication were commenced and continued indefinitely, for unclear reasons.
In 2015, he represented with inferior STEMI, having self- ceased aspirin and clopidogrel 7 months prior. CA revealed mid-vessel RCA stent thrombosis. Following RCA clot aspiration, IVUS demonstrated severe in-stent restenosis with subsequent plaque rupture. A DES was then successfully deployed.
Discussion
Very late DES thrombosis, beyond 1 year, is being increasingly described and stent specific risk factors have been identified. Cases of very, very late DES thrombosis, beyond 5 years, have been described, occurring to 7.1 years and to 12 years in bare metal stents (1, 2). Both our cases represent very, very late DES thrombosis beyond 10 years, yet with different etiologies as elucidated by IVUS. Additionally, case 1 reflects the rare scenario of recurrent episodes of VLST in the same vessel.
Case 1 demonstrates many stent specific causative risk factors for VLST: a long stented length of vessel; incomplete stent apposition with non-endothelialised stent struts, likely peri-stent inflammation and positive arterial remodeling.
Stent length is positively correlated with the rate of ST and risk of major adverse cardiovascular events (3). The stented segment in case 1 is 72 mm, putting the patient at higher risk of both outcomes.
Incomplete stent apposition (ISA) hampers endothelialisation and is an independent risk factor for ST. Positive arterial remodeling (a regional increase in the radius of the external elastic membrane) is the main cause of ISA and was demonstrated well in case 1. Animal studies show the inflammatory response to a DES (the major cause for positive remodeling) peaks after the eluted drug has been completely released (4) suggesting exposure to the stent polymer or the lack of drug elution as the cause. Registries suggest lower rates of VLST in newer generation DES, compared to older models, such as the TAXUS stents used in our cases (5). This relates to the polymer used, strut design and type of eluted drug. Additionally, resolution of jailed thrombus, insufficient neointimal hyperplasia and chronic stent recoil has also been implicated in ISA.
Stent under expansion is another risk factor for ISA and ST. In case 1, IVUS showed a vessel diameter of 3.5mm (excluding remodeled segments) with the stents originally being deployed to 2.75 and 2.5 mm.
Case 1 and case 2 demonstrated VLST at 11 years from in-stent restenosis and plaque rupture in the setting of antiplatelet discontinuation. Antiplatelet cessation is the major predictor of both early and late ST. The mean time to ST following cessation of aspirin monotherapy in first generation DES is 7 days.
Patients with observed positive remodeling in relation to a coronary stent should be considered at high risk of further ST and long term dual anti-platelet therapy should be considered. Polymer-free drug eluting stents or bio-absorbable stent scaffold, may theoretically offer a solution, however more long term efficacy data is needed given similar rates of ST after 16 months (6). These cases also illustrate the utility of IVUS, in defining pathology as well as directing definitive treatment.
Conclusion
The cases highlight the need for suspicion of VLST when patients with a prior history of PCI present with STEMI, even beyond ten years following initial PCI, especially if ST has occurred previously.
Peer-review: Internal and external
Conflict of interest: None to declare
Authorship: S.L. and M.L. equally contributed to management of case and preparation of case report.
Acknowledgement and funding: None to declare.
Informed consent of patients was obtained before procedures.
References
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||
Copyright

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
AUTHOR'S CORNER

