Persistent right atrial standstill in a patient with myasthenia gravis: A case report
CASE REPORT
Persistent right atrial standstill in a patient with myasthenia gravis: A case report
Article Summary
- DOI: 10.24969/hvt.2026.645
- CARDIOVASCULAR DISEASES
- Published: 24/04/2026
- Received: 20/03/2026
- Revised: 12/04/2026
- Accepted: 13/04/2026
- Views: 495
- Downloads: 296
- Keywords: Atrial standstill, right atrial standstill, myasthenia gravis, pacemaker implantation, conduction disorder, sino-ventricular conduction
Address for Correspondence: Bernard Abi-Saleh, Section of Electrophysiology and Pacing, Division of Cardiology, American University of Beirut, Medical Center, Beirut, Lebanon
Randa Tabbah, Division of Cardiology Notre Dame des Secours University Hospital, Byblos; Lebanese American University Medical Center, Beirut, Lebanon
Email: Bernard Abi-Saleh - Ba47@aub.edu.lb, Randa Tabbah - randa_22tabbah@hotmail.com
ORCID: Randa Tabbah - 0000-0003-2987-9568
Randa Tabbah1,2,3, Moied Al Sakan4, Bernard Abi-Saleh5
1Division of Cardiology, Holy Spirit of Kaslik University, Notre Dame de Secours University Hospital, Beirut, Lebanon
2Division of Cardiology, St John University Hospital, Lebanese American University, Beirut, Lebanon
3Division of Cardiology, Balamand University, Notre Dame du Liban University Hospital, Beirut, Lebanon
4Division of Cardiology, American University of Beirut Medical Center, Beirut, Lebanon
5Section of Electrophysiology and Pacing, Division of Cardiology, American University of
Beirut Medical Center, Beirut, Lebanon
Abstract
Objective: Atrial standstill is a rare arrhythmogenic disorder characterized by the complete absence of atrial electrical and mechanical activity. It has been described in association with structural heart disease, cardiomyopathies, and neuromuscular disorders, but persistent right atrial standstill remains exceptionally uncommon.
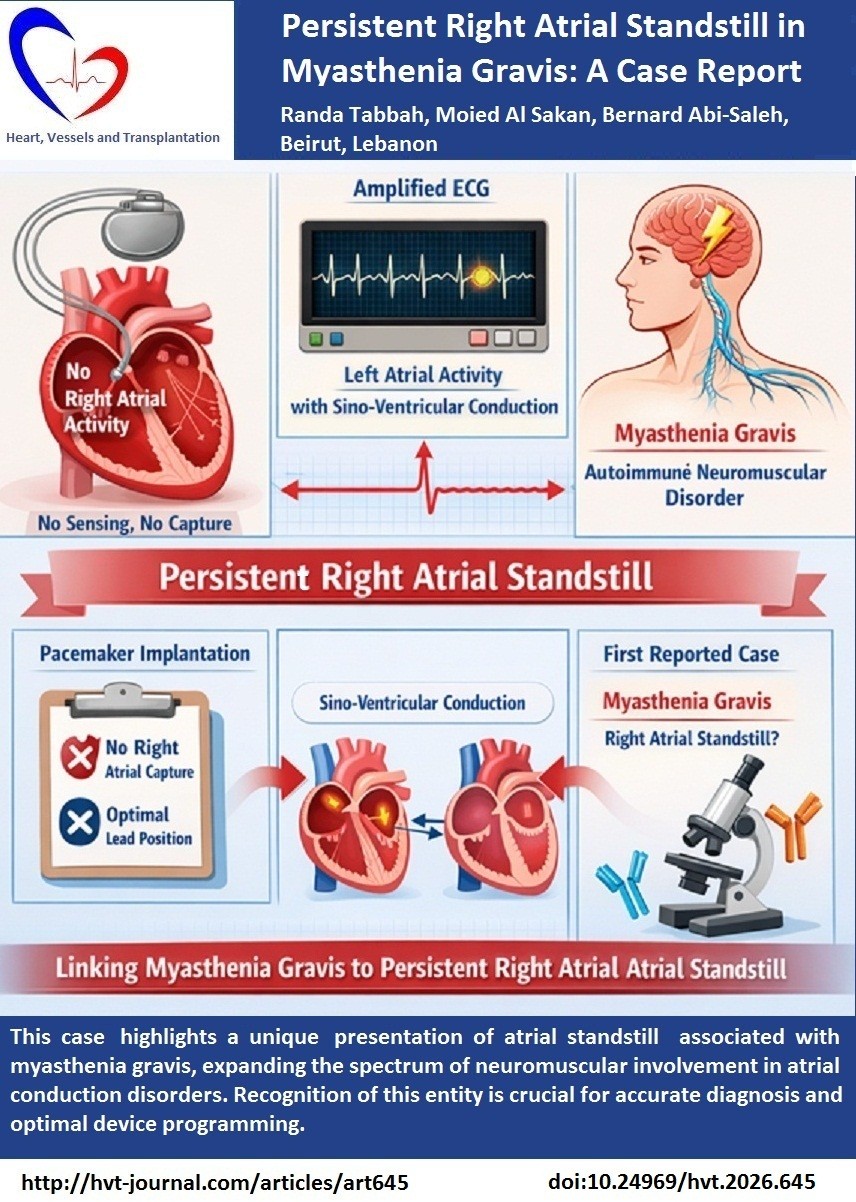
Case presentation: We report the case of a 70-year-old man with a history of myasthenia gravis, coronary artery disease, and chronic obstructive pulmonary disease who presented with tachy-brady syndrome and persistent atrial fibrillation. During pacemaker implantation, the absence of atrial capture and sensing was confirmed despite multiple attempts and appropriate lead positioning, consistent with persistent atrial standstill. Electrocardiography revealed P waves only after increasing the recording amplitude, suggesting preserved left atrial activity and probable sino-ventricular conduction. To our knowledge, this is the first reported case linking myasthenia gravis with persistent right atrial standstill.
Conclusion: This case highlights a unique presentation of atrial standstill associated with myasthenia gravis, expanding the spectrum of neuromuscular involvement in atrial conduction disorders. Recognition of this entity is crucial for accurate diagnosis and optimal device programming.
Key words: Atrial standstill, right atrial standstill, myasthenia gravis, pacemaker implantation, conduction disorder, sino-ventricular conduction

Graphical abstract
Introduction
Atrial standstill is a phenomenon characterized by the absence of both electrical and mechanical atrial activity, resulting in a complete lack of excitability. Identified causes include drugs, electrolyte abnormalities, hypoxia, myocardial infarction and neuromuscular disease.
Atrial standstill was first described by Chávez et al. in 1946 . The diagnostic criteria include the absence of P waves on electrocardiogram (ECG) and A waves on the intracardiac recordings, evidence of atrial paralysis with the absence of A waves in the jugular venous pulse, in the atrial pressure recording and on the mitral Doppler recording, and inability to stimulate the atria. It has been described in association with structural heart disease, cardiomyopathies, and neuromuscular disorders, but persistent right atrial standstill remains exceptionally uncommon.
We present as case with persistent atrial standstill in a patient with miastenia gravis (MG) undergoing pacemaker implantation.
Case report
A 70-year-old man with a history of remote alcohol dependency, former smoker (1.5 packs per day), MG, chronic obstructive pulmonary disease (COPD) and coronary artery disease (CAD) with subtotally occluded mid left anterior descending artery treated with angioplasty and persistent atrial fibrillation with rapid ventricular response. In addition, he had pronounced bradycardia on low-dose of beta-blockers.
Written consent from the patient was obtained for all procedures and treatment.
A cardioversion was attempted but failed to restore sinus rhythm despite multiple shocks. Two weeks later, he presented to the clinic in a regular rhythm at 80 beats per minute. ECG showed a regular narrow complex rhythm, and then a double amplitude recording uncovered the P waves (Fig. 1). Given his tachy-brady syndrome, a decision was taken to implant a pacemaker.
His current home medications included budesonide, ipratropium bromide as needed for his COPD, furosemide 40 mg one tablet daily, atorvastatin 40mg at night, bisoprolol 2.5 mg in the morning, and rivaroxaban 20 mg daily. His echocardiogram revealed a dilated left ventricle with a mild left ventricular dysfunction, with an estimated Left ventricular ejection fraction of 40-44%, global hypokinesis, high left ventricular end-diastolic pressure and severe bi-atrial enlargement; no right ventricular dysfunction was noticed, nor any valvular disease.
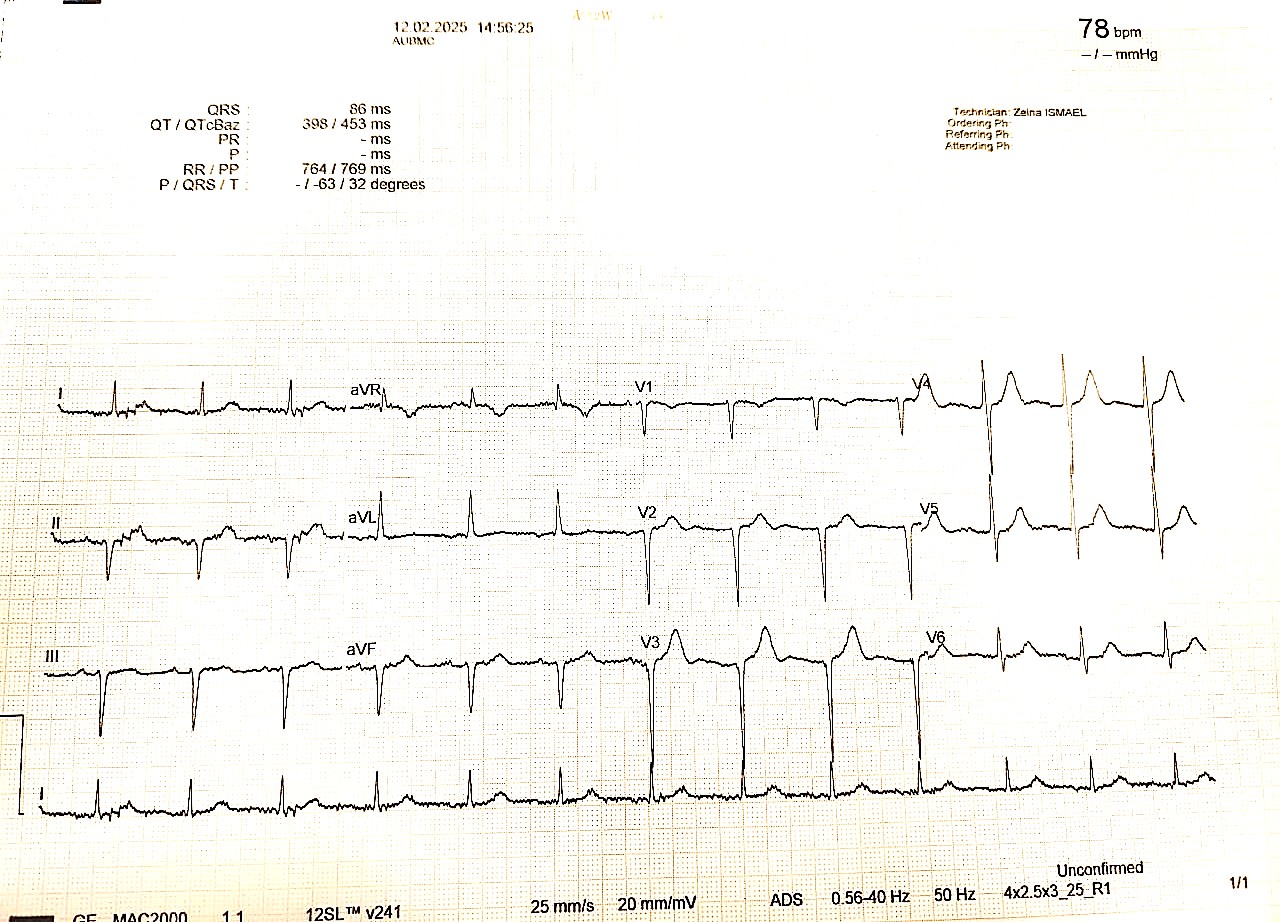
Figure 1. Visible P waves after increasing the amplitude on the ECG machine
ECG –electrocardiogram
A multidisciplinary decision to implant a dual chamber pacemaker with a conduction system pacing (CSP), was taken.
During the procedure, A 3830-conduction system lead was placed in the mid-septum with confirmed capture of the left bundle branch (left ventricular activation time, LVAT of 70 msec and an interpeak distance of 45 msec).
An appropriate capture and sensing threshold of the ventricular lead was demonstrated. Then an atrial lead was implanted, but no sensing of an atrial activity was detected despite multiple positions in several areas of the right atrium and no capture of atrial activity, even though the lead was tested in the ventricle and had appropriate impedance. Fluoroscopy showed no right atrial contraction.
Given this fact, the pacemaker settings were programmed to a VVIR mode.
ECG was done after the procedure (Fig. 2).
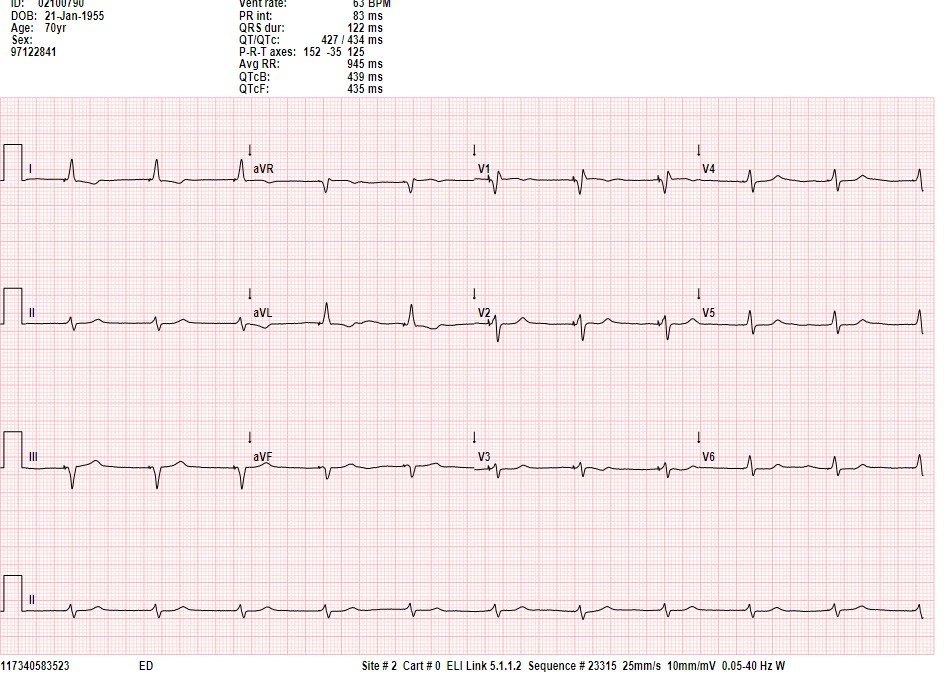
Figure 2. ECG after implantation of a CSP lead
CSP – conduction system pacing, ECG –electrocardiogram
The patient was discharged on medical therapy to improve left ventricular function and symptoms, including empagliflozin and an aldosterone antagonist and kept on direct oral anticoagulation.
After a follow-up at one month, testing of the device continued to show no atrial activity, no P wave noticed and no right atrial capture.
Discussion:
Atrial standstill is a rare phenomenon. A temporary atrial standstill can sometimes result from heart disease or cardioversion. A persistent right atrial standstill is very rare.
The patient described in this case presented with a persistent right atrial standstill, whereby no right atrial activity, nor atrial capture were seen during pacemaker implantation and on follow-up. On the other hand, the ECG presented an atrial activity P wave (Fig. 1). Atrial activity was noticed after increasing the amplitude. Therefore, the ECG suggests a sinus rhythm rather than an ectopic atrial rhythm or junctional rhythm. This will imply that the conduction from the sinus node to the left atrium is intact despite the absence of right atrial activity and increases the possibility of sinoventricular conduction along specialized pathways like Bachmann bundle. Thus, this activity probably originates from a left atrial contraction. Only one previous case in the literature reported a persistent right atrial standstill but with no associated heart disease or neuromuscular disease and a second one with persistent Isolated right atrial standstill associated with left atrial tachycardia .
Most reported cases of atrial standstill have been associated with clinical conditions, including myocarditis, muscular dystrophies, Emery-Dreifuss muscular dystrophy , spinal muscular atrophy, and infiltrative and dilated cardiomyopathies . Familial form associated with Na+-channel mutation has also been described (6-8).
The described patient had a known history of MG known as a neuromuscular disease. No cases were reported in the literature associating MG and atrial standstill before. Moreover, myasthenia gravis is an autoimmune neuromuscular disorder with cardiac involvement. Despite atrial standstill was seen before in other neuromuscular diseases patients with MG presented with arrhythmia, sick sinus syndrome, and heart block in those patients as described by Nag et al (9). The autoimmune response can lead in certain cases to conduction disturbance and, theoretically, to atrial standstill.
This is a unique case describing a persistent right atrial standstill diagnosed during a pacemaker implantation procedure. It’s the first case where atrial standstill is described in MG patient.
Conclusion
This case highlights a unique presentation of atrial standstill associated with myasthenia gravis, expanding the spectrum of neuromuscular involvement in atrial conduction disorders. Recognition of this entity is crucial for accurate diagnosis and optimal device programming.
Ethics: Written consent from the patient was obtained for all procedures and treatment. The study was performed in frame of Helsinki 2024 agreement for human studies.
Peer-review: External and Internal
Conflict of interest: None to declare
Authorship: R.T., M.A.S., B.A-S. equally contribute to the management of patients and preparation of case report, thus fulfilled all authorship criteria
Acknowledgements and Funding: None to declare.
Statement on A.I.-assisted technologies use: Author stated they did not use artificial intelligence (A.I.) tools for preparation of manuscript
Data and material availability: Does not apply
References
| 1.Woolliscroft J, Tuna N. Permanent atrial standstill: the clinical spectrum. Am J Cardiol 1982; 49: 2037-41. doi:10.1016/0002-9149(82)90226-0 https://doi.org/10.1016/0002-9149(82)90226-0 PMid:7081083 |
||||
| 2.Jorat MV, Nikoo MH, Yousefi A. Persistent isolated right atrial standstill associated with left atrial tachycardia. Res Cardiovasc Med 2014;3: e25173. doi:10.5812/cardiovascmed.25173 https://doi.org/10.5812/cardiovascmed.25173 PMid:25785252 PMCid:PMC4347757 |
||||
| 3.Harley A. Persistent right atrial standstill. Br Heart J 1976; 38: 646-9. doi:10.1136/hrt.38.6.646 https://doi.org/10.1136/hrt.38.6.646 PMid:132178 PMCid:PMC483049 |
||||
| 4.Rodríguez-Torres D, Jiménez-Jáimez J, Macías-Ruiz R, Álvarez-López M, Tercedor L. Cardiac manifestations of neuromuscular disease. Rev Esp Cardiol (Engl Ed) 2018; 71: 580-2. doi:10.1016/j.rec.2017.03.033 https://doi.org/10.1016/j.rec.2017.03.033 PMid:28601412 |
||||
| 5.Zhang Y, Zhang Y, Ren M, XXX, XXX, XXX, et al. Atrial standstill associated with lamin A/C mutation: A case report. SAGE Open Med Case Rep 2023; 11: 2050313X231179810. doi:10.1177/2050313X231179810 https://doi.org/10.1177/2050313X231179810 PMid:37425136 PMCid:PMC10328153 |
||||
| 6.Feng JY, Vashistha K, Chatterjee NA, Mansour MC, Heist EK, Maan A. Atrial standstill due to eosinophilic myocarditis and drug reaction with eosinophilia and systemic symptoms. Heart Rhythm Case Rep 2024; 11: 229-33. doi:10.1016/j.hrcr.2024.11.019 https://doi.org/10.1016/j.hrcr.2024.11.019 PMid:40182941 PMCid:PMC11962991 |
||||
| 7.Howard TS, Chiang DY, Ceresnak SR, XXX, XXX, XXX, et al. Atrial standstill in the pediatric population: A multi-institution collaboration. JACC Clin Electrophysiol 2023; 9: 57-69. doi:10.1016/j.jacep.2022.08.022 https://doi.org/10.1016/j.jacep.2022.08.022 PMid:36435694 PMCid:PMC12998638 |
||||
| 8.Bozkurt B, Colvin M, Cook J, XXX, XXX, XXX, et al. Current diagnostic and treatment strategies for specific dilated cardiomyopathies: A scientific statement from the American Heart Association. Circulation 2016; 134: e579-e646. doi:10.1161/CIR.0000000000000455 https://doi.org/10.1161/CIR.0000000000000455 |
||||
| 9.Nag DS, Chatterjee A, Mahanty PR, Sam M, Bharadwaj MK. Perioperative cardiac risks in myasthenia gravis. World J Clin Cases 2024; 12: 2147-50. doi:10.12998/wjcc.v12.i13.2147 https://doi.org/10.12998/wjcc.v12.i13.2147 PMid:38808348 PMCid:PMC11129138 |
||||
Copyright

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
AUTHOR'S CORNER

