The role of hemodynamic factors in arterial stiffness development during the early stages of peripheral vascular remodeling: A single-center observational cross-sectional study
ORIGINAL RESEARCH ARTICLE
The role of hemodynamic factors in arterial stiffness development during the early stages of peripheral vascular remodeling: A single-center observational cross-sectional study
Article Summary
- DOI: 10.24969/hvt.2026.646
- CARDIOVASCULAR DISEASES
- Published: 30/04/2026
- Received: 07/03/2026
- Revised: 07/04/2026
- Accepted: 07/04/2026
- Views: 13
- Downloads: 10
- Keywords: Arterial stiffness, peripheral arterial remodeling, ultrasonography, photoplethysmography, pulse pressure, augmentation index
Address for Correspondence: Gulbarchyn Zh. Suranova, Department of Therapy-1, Medical Faculty, Kyrgyz-Russian Slavic University named after B.N. Yeltsin, Kievskaya St., 44, Bishkek 720000, Kyrgyz Republic
Email: czhsuranova@gmail.com Mobile: +996 772 665 006
ORCID: Gulbarchyn Zh. Suranova: 0000-0002-9458-487; Uuulzhan A. Alieva: 0009-0008-5009-4267; Aruuke A.Abdumanapova: 0009-0000-1184-3693; Samatbek K. Abdrahmanov: 0009-0006-0889-6130
Gulbarchyn Zh. Suranova, Uuulzhan A. Alieva, Aruuke A.Abdumanapova, Samatbek K. Abdrahmanov
Department of Therapy-1 (Pediatrics and Dentistry), Medical Faculty, Interstate Educational Institution of Higher Education Kyrgyz-Russian Slavic University named after the First President of the Russian Federation B.N. Yeltsin, Bishkek, Kyrgyz Republic
Abstract
Objective: Peripheral vascular remodeling at early stages is associated with increased risk of cardiovascular complications. Arterial stiffness is a key marker of vascular aging and may be determined by both chronological age and local hemodynamic loads.
We aimed to study the structure of determinants of arterial stiffness in individuals with and without early ultrasound signs of peripheral arterial remodeling.
Methods: A single-center cross-sectional observational study included 90 adults (27–84 years), of whom 19 were male (21.1%) and 71 were female (78.9%), divided into a control group without signs of remodeling (n = 37, 41.1%) and a study group with early remodeling (n = 53, 58.9%). Arterial stiffness was assessed using photoplethysmography (adjusted stiffness index — ASI). The structure of ASI determinants was analyzed using hierarchical linear regression.
Results: Groups were comparable by age, anthropometric parameters, and blood pressure (BP) levels (systolic BP, diastolic BP, pulse pressure, mean arterial pressure and central systolic BP (all p>0.05). Heart rate was significantly higher in the early remodeling group (p = 0.025). In the study group, pulse pressure and augmentation index were significantly associated with ASI (β = 0.386; p = 0.010 and β = 0.363; p = 0.014, respectively), whereas age was not a significant predictor (β = −0.104; p = 0.497). In the control group, hemodynamic factors did not reach statistical significance (pulse pressure: p = 0.341; augmentation index: p = 0.060), reflecting preserved vascular elasticity.
Conclusion: During early peripheral vascular remodeling, local hemodynamic loads — measured as pulse pressure and augmentation index — play a leading role in arterial stiffness formation, whereas in the absence of structural changes no association between ASI and hemodynamic parameters was found. Prospective studies with larger cohorts are needed to confirm these findings and assess their prognostic significance.
Key words: Arterial stiffness, peripheral arterial remodeling, ultrasonography, photoplethysmography, pulse pressure, augmentation index

Graphical abstract
Introduction
Peripheral vascular changes are an important component of cardiovascular pathology and are associated with increased risk of adverse clinical outcomes (1, 2). Increased arterial stiffness is considered an independent predictor of cardiovascular morbidity and mortality, reflecting processes of vascular aging and remodeling (1, 2).
Vascular remodeling represents a continuum of structural and functional changes in the arterial wall in response to hemodynamic and metabolic stimuli (1, 3, 4). Early stages are characterized by functional alterations, including endothelial dysfunction, changes in smooth muscle cell tone, and disturbances in extracellular matrix composition, without significant morphological changes (4, 5). Late stages involve progressive intimal thickening, formation of atherosclerotic plaques, and calcification, leading to irreversible structural alterations (1, 3). Understanding these distinct phases is essential for identifying appropriate intervention targets and preventing progression to advanced atherosclerotic disease.
Traditionally, arterial stiffness assessment is based on pulse wave velocity (PWV) and augmentation index (AI), recognized as validated markers of central hemodynamics (1, 6). In recent years, methods based on photoplethysmographic (PPG) waveform analysis have become widely used, allowing calculation of the adjusted stiffness index (ASI), age index (AGI), and heart rate-normalized augmentation index (AIP₇₅) (5–9). Studies have shown that PPG-derived ASI correlates with classical stiffness measures and has prognostic significance in population studies (2, 10).
At early stages of vascular remodeling, changes are predominantly functional, manifesting as disturbances in reflected wave parameters and local hemodynamics without pronounced atherosclerotic lesions. However, the structure of determinants of arterial stiffness in the presence of early ultrasound signs of peripheral remodeling remains insufficiently studied.
Intima-media thickness (IMT) of the femoral artery is used as a research indicator of early subclinical atherosclerosis. Although international guidelines (11) do not include femoral artery IMT in standard clinical markers, contemporary studies demonstrate its significance for assessing structural vascular wall remodeling and detecting early functional changes before atherosclerotic plaque formation (3, 7). Femoral artery IMT was selected for this study as it represents peripheral vascular bed affected by early remodeling, while carotid IMT assessment was beyond the scope of this hemodynamic-focused study (3). This approach is justified by the fact that peripheral arteries, including femoral, demonstrate earlier functional changes compared to carotid arteries in the initial stages of atherosclerotic process (3). Detection of early peripheral remodeling is clinically important as it identifies patients at elevated cardiovascular risk before the development of hemodynamically significant stenosis or overt atherosclerotic plaques, offering a window for preventive intervention.
Particular interest lies in analyzing the contribution of chronological age and current hemodynamic load to stiffness indicator formation. In population studies, age is considered the leading factor of vascular aging (2, 10); however, with structural changes in the vascular wall, the role of pulse arterial pressure may increase.
Our objective was to study the structure of determinants of arterial stiffness and their differences in individuals with and without early ultrasound signs of peripheral arterial remodeling.
Methods
Study design and population
A single-center cross-sectional observational study was conducted with group formation based on ultrasound assessment of peripheral arteries. Data collection was conducted in 2025 following acquisition of the AngioCode-322 photoplethysmography system. The study was reported in accordance with the STROBE statement for observational studies.
The study included 90 adult participants (27–84 years), mean age 60.9 (1.2) years. Of these, 19 were male (21.1%) and 71 were female (78.9%). Recruitment was conducted by consecutive sampling among individuals undergoing clinical and instrumental examination as part of planned cardiovascular screening at a regional outpatient facility. Distribution of patients in groups was based on duplex ultrasound scanning data of lower limb arteries. Two groups were formed:
Control group (n = 37, 41.1%) - individuals without signs of peripheral arterial remodeling (IMT < 1.0 mm, no atherosclerotic plaques, no hemodynamically significant stenoses);
Early remodeling group (n = 53, 58.9%) - individuals with early peripheral arterial remodeling (IMT 1.0–1.4 mm without atherosclerotic plaques or hemodynamically significant stenoses of lower limb arteries), corresponding to the preclinical stage of peripheral arterial remodeling (3).
Arterial hypertension was not a criterion for group distribution: hypertensive patients were present in both groups.
Inclusion criteria were: age ≥ 18 years; signed informed consent; ability to undergo photoplethysmographic examination and duplex ultrasound scanning of lower limb arteries. Exclusion criteria were as following: hemodynamically significant stenoses or occlusions; atherosclerotic plaques (IMT ≥ 1.5 mm); acute cardiovascular events within 6 months; chronic heart failure III–IV functional class; clinically significant arrhythmias; systemic inflammatory diseases in exacerbation; diabetes mellitus; medications substantially altering pulse wave parameters; technical inability to adequately visualize arteries.
The study was performed in accordance with the principles of the Declaration of Helsinki and approved by the Ethics Committee of the Scientific and Production Association "Preventive Medicine" of the Ministry of Health of the Kyrgyz Republic (conclusion No. 7 dated November 16, 2017). All participants signed informed consent.
Anthropometric and clinical variables
We collected demographic (age and sex), anthropometric (height and weight), hemodynamic (blood pressure and heart rate) and history of arterial hypertension data. Height and body weight were measured by standard method with calculation of body mass index (BMI, kg/m²). Blood pressure was measured in sitting position after 5-minute rest; the mean of two consecutive measurements was used. Systolic blood pressure (SBP), diastolic blood pressure (DBP), pulse pressure (PP), mean arterial pressure (MAP), and heart rate (HR) were recorded. Data on smoking status and family history of atherosclerotic cardiovascular diseases (ASCVD) were incompletely documented in medical records and therefore not included in the analysis.
Predictor variable: Ultrasound Assessment of Peripheral Arteries
Intima-media thickness (IMT) of the common femoral artery was measured using the Mindray DC-60 ultrasound system (Mindray Bio-Medical Electronics Co., Ltd., Shenzhen, China) equipped with a 7.5–12 MHz linear array transducer. Measurements were performed in B-mode with color Doppler mapping in standard segments, three times with result averaging.
Early peripheral arterial remodeling was defined as increased intima-media thickness (IMT 1.0–1.4 mm) without hemodynamically significant stenosis or formed atherosclerotic plaques, corresponding to the preclinical stage of structural vascular wall changes (3). Atherosclerosis was defined as the presence of focal lesions with IMT ≥ 1.5 mm or plaque formation.
Group allocation was based on IMT values: control group (IMT < 1.0 mm) and early remodeling group (IMT 1.0–1.4 mm without plaques).
Outcome variables: Photoplethysmography evaluation of vascular function
Vascular function assessment was performed using the AngioCode-322 PPG system (AngioCode LLC, Moscow, Russia; software version 3.2.1). Signal was registered at the finger after 10-minute adaptation rest. The following parameters were analyzed:
• Adjusted stiffness index (ASI, m/s) — main study indicator. ASI is a validated PPG-derived indicator of arterial stiffness, reflecting the velocity of pulse wave propagation in the arterial tree (7, 12);
• Augmentation index normalized to heart rate 75 bpm (AIP₇₅, %);
• Resistance index (RI);
• Age index (AGI);
• Time between systolic peak and reflected wave (dTpp, ms);
• Vascular age (years) — the estimated biological age of the arterial system based on PPG waveform morphology and ASI values compared to normative population age curves embedded in the device software.
ASI was calculated automatically with correction for height and heart rate. Before measurements, the device was calibrated against peripheral systolic and diastolic pressure measured at the brachial artery.
Central hemodynamics assessment
Central (aortic) SBP was estimated non-invasively using the AngioCode-322 device via radial artery applanation tonometry, with subsequent mathematical derivation of aortic pressure using a generalized transfer function, consistent with the methodology described in reference (4).
Statistical Analysis
Data processing was conducted using SPSS Statistics 16.0 (IBM Corp., Armonk, NY, USA). Quantitative parameters are presented as median (Me) and interquartile range (Q₁–Q₃). Normality was tested by Shapiro–Wilk test. For non-normal distribution, Mann–Whitney U test was used for group comparison. Categorical variables were compared using Chi-square test.
Correlation analysis was performed using Spearman's coefficient (ρ). To assess the contribution of age and pulse pressure (PP) to ASI variability, hierarchical linear regression was applied: Model 1 — age; Model 2 — age + PP; Model 3 — age + PP + AIP₇₅. Multicollinearity was assessed using variance inflation factor (VIF; critical value > 5). Statistical significance was accepted at p < 0.05. Statistical reporting was performed in accordance with SAMPL guidelines.
Results
Baseline Characteristics
Table 1 presents the baseline characteristics of the study participants. The two groups were comparable in age (p = 0.974), anthropometric parameters (BMI: p = 0.912), and blood pressure levels (SBP: p = 0.864; DBP: p = 0.140; PP: p = 0.253; MAP: p = 0.511; central SBP: p = 0.810) (Table 1). Sex distribution showed a numerical difference (p = 0.225). The prevalence of arterial hypertension did not differ between groups (p = 0.354). HR was significantly higher in the early remodeling group (p = 0.025), which is relevant for interpretation of AIP₇₅ normalized to 75 bpm.
|
Table 1. Clinical, anthropometric, and hemodynamic characteristics of examined individuals |
|||
|---|---|---|---|
|
Variables |
Control group (n = 37, 41.1%) |
Early remodeling group (n = 53, 58.9%) |
p |
|
Demographics |
|||
|
Age, years |
62.0 (53.0–70.0) |
63.0 (56.0–67.0) |
0.974 |
|
Male sex, n (%) |
5 (13.5%) |
14 (26.4%) |
0.225* |
|
Female sex, n (%) |
32 (86.5%) |
39 (73.6%) |
— |
|
Anthropometrics |
|||
|
Height, cm |
158.0(155.0–165.0) |
160.0 (153.0–166.0) |
0.493 |
|
Body weight, kg |
75.0 (69.0–82.0) |
75.0 (69.0–84.0) |
0.879 |
|
BMI, kg/m² |
30.1 (26.0–32.5) |
29.6 (25.4–32.5) |
0.912 |
|
Hemodynamics |
|||
|
SBP, mmHg |
121.0 (120.0–142.5) |
126.0 (116.8–144.2) |
0.864 |
|
DBP, mmHg |
80.0 (73.0–81.0) |
76.5 (68.8–81.2) |
0.140 |
|
PP, mmHg |
49.0 (40.0–56.5) |
54.0 (42.0–66.8) |
0.253 |
|
MAP, mmHg |
93.3 (90.2–104.0) |
93.8 (86.5–100.0) |
0.511 |
|
Central SBP, mmHg |
120.0 (119.0–144.0) |
125.0 (114.0–146.8) |
0.810 |
|
Heart rate, bpm |
73.0 (69.0–81.0) |
81.0 (73.2–86.0) |
0.025 |
|
CV Risk Factors |
|||
|
Arterial hypertension, n (%) |
17 (45.9%) |
18 (34.0%) |
0.354* |
|
Data presented as Median (Q1–Q3) Mann–Whitney U test and *Chi-square test BMI - body mass index, CV-cardiovascular, DBP – diastolic blood pressure, MAP – mean arterial pressure, PP – pulse pressure, SBP – systolic blood pressure |
|||
Photoplethysmographic parameters
Table 2 presents PPG-derived vascular parameters for both groups. ASI was significantly higher in the early remodeling group (p = 0.022), and dTpp was significantly shorter (p = 0.008), indicating greater arterial stiffness and altered reflected wave timing in the early remodeling group. Other PPG parameters (AIP₇₅, AGI, RI, vascular age) did not differ significantly between groups.![]()
|
Table 2. Photoplethysmographic parameters of vascular function in control and early remodeling groups |
|||
|---|---|---|---|
|
Variables |
Control group (n = 37) |
Early remodeling group (n = 53) |
p |
|
Vascular age, years |
57.0 (45.2–67.0) |
61.0 (50.2–67.5) |
0.336 |
|
ASI, m/s |
8.4 (7.4–9.6) |
9.1 (7.8–11.3) |
0.022 |
|
AIP₇₅, % |
17.2 (5.5–26.0) |
12.4 (6.5–25.2) |
0.505 |
|
AGI |
−0.4 (−0.7–−0.1) |
−0.3 (−0.5–−0.1) |
0.174 |
|
dTpp, ms |
98.0 (87.2–109.8) |
86.0 (69.5–100.5) |
0.008 |
|
RI, % |
33.9 (22.7–51.7) |
36.2 (30.4–54.1) |
0.282 |
|
Data presented as Median (Q1–Q3), Mann–Whitney U test AGI - age index, AIP₇₅ - augmentation index normalized to heart rate 75 bpm, ASI - adjusted stiffness index, dTpp - time between systolic peak and reflected wave, RI - resistance index |
|||
Hemodynamic parameters
Comparison of blood pressure parameters between groups revealed no statistically significant differences in systolic, diastolic, pulse, mean arterial, or central systolic pressures (all p > 0.05), despite numerically higher PP in the early remodeling group (54.0 vs 49.0 mmHg) (Fig. 1).
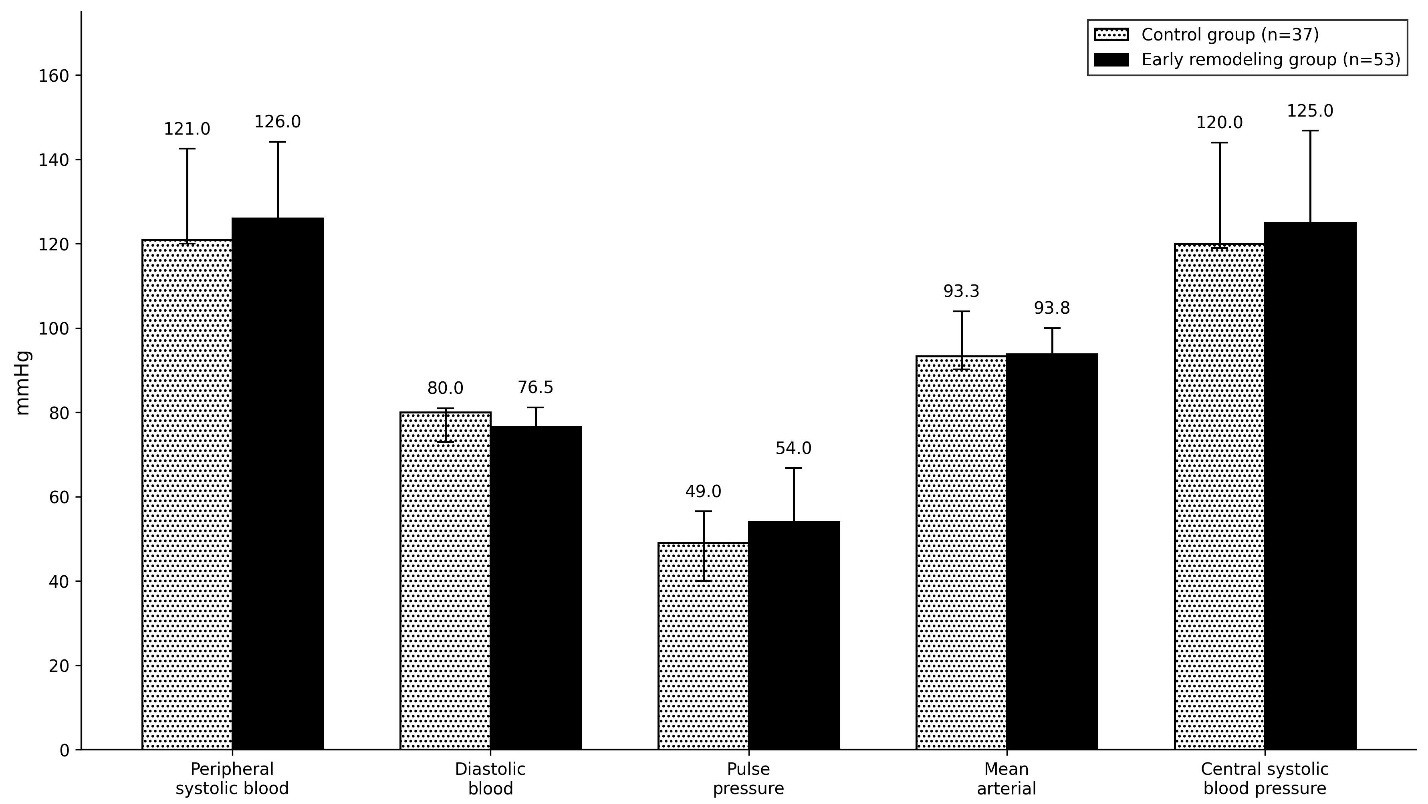
Figure 1. Hemodynamic parameters in control and early remodeling groups
Data are presented as median and interquartile range (Me (Q1–Q3)), (p > 0.05)
Correlation analysis
In the control group, a moderate positive correlation was found between chronological and vascular age (ρ = 0.528; p < 0.01). In the early remodeling group, this association was statistically non-significant (ρ = 0.103; p > 0.05), indicating altered age-related determination of vascular parameters. The association between height and ASI had opposite direction in groups: moderate negative in the control group (ρ = −0.401; p < 0.05) and moderate positive in the early remodeling group (ρ = 0.420; p < 0.01) (Table 3).
|
Table 3. Correlation between age, anthropometric characteristics and vascular parameters |
||
|---|---|---|
|
Variables |
Control group (ρ, p) |
Early remodeling group (ρ, p) |
|
Age — Vascular age |
0.528 (0.002) |
0.103 (0.463) |
|
Height — ASI |
−0.401 (0.015) |
0.420 (0.002) |
|
Vascular age — AGI |
0.864 (<0.001) |
0.965 (<0.001) |
|
ASI — dTpp |
−0.908 (<0.001) |
−0.937 (<0.001) |
|
Peripheral SBP — Central SBP |
0.892 (<0.001) |
0.977 (<0.001) |
|
ρ = Spearman's rank correlation coefficient AGI - age index, ASI - adjusted stiffness index, dTpp - time between systolic peak and reflected wave, SBP - systolic blood pressure, VA - vascular age |
||
Regression analysis of determinants of increase arterial stiffness
To assess the contribution of individual parameters to ASI explanation, hierarchical linear regression was performed. Predictors were entered sequentially: first age (Model 1), then PP (Model 2), and finally AIP₇₅ (Model 3). Height was not included despite significant correlation with ASI, as ASI is already corrected for height by the AngioCode-322 device software (version 3.2.1), which would create statistical redundancy.
In the control group, age was not a significant predictor of ASI (R² = 0.019; β = 0.137; p = 0.512). Adding pulse pressure and augmentation index slightly increased explained variance, with predictors remaining statistically non-significant (Table 4).
|
Table 4. Hierarchical linear regression analysis of ASI determinants by group |
|||||||
|---|---|---|---|---|---|---|---|
|
Model |
Predictor |
R² (Control) |
ΔR² (Control) |
p (Control) |
R² (Remodeling) |
ΔR² (Remodeling) |
p (Remodeling) |
|
1 |
Age |
0.019 |
0.019 |
0.512 |
0.092 |
0.092 |
0.042 |
|
2 |
Age + PP |
0.034 |
0.015 |
0.341 |
0.157 |
0.064 |
0.010 |
|
3 |
Age + PP + AIP₇₅ |
0.099 |
0.065 |
0.060 |
0.272 |
0.115 |
0.014 |
|
R²- coefficient of determination; ΔR² - increase in explained variance AIP₇₅ = augmentation index at heart rate 75 bpm, ASI – arterial stiffness index, PP - pulse pressure |
|||||||
In the early remodeling group, pulse pressure was a significant predictor of ASI in Model 2 (β = 0.386; p = 0.010). With addition of AIP₇₅, model explanatory power increased to R² = 0.272, and AIP₇₅ also became an independent significant predictor (β = 0.363; p = 0.014). Age remained statistically non-significant in all models (Table 5).
|
Table 5. Standardized predictor coefficients for ASI |
||||||
|---|---|---|---|---|---|---|
|
Predictor |
β (Control) |
p |
VIF |
β (Remodeling) |
p |
VIF |
|
Age |
0.137 |
0.512 |
1.00 |
−0.104 |
0.497 |
1.00 |
|
PP |
0.204 |
0.341 |
1.03 |
0.386 |
0.010 |
1.03 |
|
AIP₇₅ |
0.541 |
0.060 |
1.96 |
0.363 |
0.014 |
1.14 |
|
β - standardized regression coefficient, VIF - variance inflation factor AIP₇₅ = augmentation index at heart rate 75 bpm, ASI – arterial stiffness index, PP - pulse pressure |
||||||
Discussion
The present study revealed that early peripheral vascular remodeling is accompanied by restructuring of arterial stiffness determinants. Although no intergroup differences in absolute blood pressure levels were found, significant differences were observed in the structure of relationships: in the early remodeling group, ASI was significantly associated with hemodynamic parameters (pulse pressure and AIP₇₅), whereas no such associations were found in the control group.
Comparison with published studies
Szołtysek-Bołdys et al. (10) demonstrated that PPG-derived stiffness parameters differ between patients with and without arterial hypertension and atherosclerosis, consistent with our finding that structural changes alter the hemodynamic determinants of stiffness. Chen et al. (2) showed that PPG-derived ASI carries greater cardiovascular prevention value in non-elderly populations, suggesting that ASI is particularly informative when used in conjunction with functional hemodynamic assessment — as demonstrated in our study. Our finding that ASI was significantly higher in the early remodeling group (9.1 vs 8.4 m/s, p = 0.022) is consistent with Szołtysek-Bołdys et al. (10), who reported elevated PPG stiffness parameters in patients with subclinical atherosclerosis. The observed dissociation between chronological age and ASI in the remodeling group (β = −0.104, p = 0.497) aligns with the concept of accelerated vascular aging independent of biological age, as described by Chen et al. (2) in non-elderly populations with increased cardiovascular risk.
This restructuring of indicator relationships reflects pathophysiological mechanisms of arterial stiffness. Normally, arterial stiffness is determined by extracellular matrix structure, elastin-to-collagen ratio, endothelial function, and smooth muscle cell tone. Increased PP enhances mechanical load on the vascular wall, stimulating extracellular matrix remodeling, collagen deposition, and progressive loss of arterial elasticity, which amplifies reflected wave and augmentation index (12).
Correlation analysis showed that age-related influence on vascular age is attenuated during early remodeling, while ASI becomes sensitive to local hemodynamic load. This emphasizes the need to account for the pathophysiological role of PP and reflected wave in assessing early vascular function changes (5, 10).
Interestingly, the association between height and ASI had opposite directions in groups. In the control group, greater height was associated with lower stiffness; consistent with physiology (longer arterial tree reduces wave reflection). In the remodeling group, this association inverted, possibly reflecting impaired arterial elasticity with structural changes.
Significantly shorter dTpp in the early remodeling group (86.0 vs 98.0 ms, p = 0.008) is consistent with earlier wave reflection, a hallmark of increased arterial stiffness, and correlates with the higher ASI observed in that group.
Study limitations
This study has several limitations that should be considered when interpreting the results: (1) relatively homogeneous study population from a single region, which may limit generalizability; (2) absence of a healthy population control group without any cardiovascular risk factors; (3) relatively small sample size (n = 90), which reduces statistical power; (4) sex distribution differed between groups (p = 0.225), with higher proportion of males in the early remodeling group (26.4% vs 13.5%), and lack of stratification by sex and other individual risk factors; (5) data on smoking status and family history of ASCVD were not systematically collected; (6) cross-sectional design, which precludes causal inference and assessment of longitudinal cardiovascular outcomes; (7) ethical approval was obtained in 2017, but data collection was performed in 2025 following acquisition of specialized photoplethysmography equipment; this delay reflects resource constraints and conduct of other research projects during the intervening period; (8) the study population was predominantly female (78.9%), reflecting higher healthcare utilization among women in this age group; this may limit generalizability of findings to male populations.
Conclusion
Early peripheral vascular remodeling is accompanied by restructuring of arterial stiffness determinants. In the absence of structural changes, ASI is not associated with hemodynamic factors; whereas with initial ultrasound signs of remodeling, local hemodynamic loads— primarily pulse pressure and heart rate-normalized augmentation index (AIP₇₅) — acquire key importance. Chronological age was not a significant predictor of ASI in either group.
This reflects not so much an increase in absolute stiffness level as a change in its formation mechanisms at early remodeling stages. Integration of functional (PPG) and structural (duplex ultrasound) markers is necessary for comprehensive vascular function assessment and cardiovascular risk stratification. Prospective studies with larger cohorts are needed to confirm the observed patterns and assess their prognostic significance.
Ethics: All participants signed informed consent. The study was performed in accordance with the principles of the Declaration of Helsinki 2024 and approved by the Ethics Committee of the Scientific and Production Association "Preventive Medicine" of the Ministry of Health of the Kyrgyz Republic (conclusion No. 7 dated November 16, 2017)
Peer-review: External and internal
Conflict of interest: None to declare
Authorship: G.Zh.S. — data collection, data processing, methodology development, data analysis, manuscript preparation; U.A.A — data collection, data processing, text editing; A.A.A.— data collection, data visualization, results verification, discussion participation; S.K.A. — statistical analysis consultation, data interpretation, manuscript preparation participation. All authors critically read and approved revised version for publication, equally contributed to the preparation of manuscript and thus fulfilled authorship criteria
Acknowledgements and Funding: None to declare.
Statement on A.I.-assisted technologies use: Author stated they did not use artificial intelligence (A.I.) tools for preparation of the manuscript
Data and material availability: Available upon request from the authors. Any sharing should follow principles of fair use with acknowledgment of source and /or collaboration.
References
| 1.Sultan SR. The association of arterial pulse wave velocity with internal carotid artery blood flow in healthy subjects: A pilot study. Artery Res 2024; 30: 7. https://doi.org/10.1007/s44200-024-00053-9 |
||||
| 2.Chen H, Fan F, Ye Z, Zhang L, Li Y, Wang J, et al. Photoplethysmography-derived arterial stiffness index delivered greater cardiovascular prevention value to non-elderly: UK Biobank retrospective study. J Clin Hypertens 2025; 27: e70058. doi:10.1111/jch.70058 https://doi.org/10.1111/jch.70058 PMid:40346852 PMCid:PMC12064935 |
||||
| 3.Szabóová E, Lisovszki A, Rajnič A, et al. Subclinical atherosclerosis progression in low-risk, middle-aged adults: Carotid leads femoral in IMT increase but not in plaque formation. J Cardiovasc Dev Dis 2024; 11: 271. doi:10.3390/jcdd11090271 https://doi.org/10.3390/jcdd11090271 PMid:39330329 PMCid:PMC11432545 |
||||
| 4.Lin PK, Davis GE. Extracellular matrix remodeling in vascular disease: defining its regulators and pathological influence. Arterioscler Thromb Vasc Biol 2023; 43: 1599-616. doi:10.1161/ATVBAHA.123.318237 https://doi.org/10.1161/ATVBAHA.123.318237 PMid:37409533 PMCid:PMC10527588 |
||||
| 5.Karimpour P, May JM, Kyriacou PA. Photoplethysmography for the assessment of arterial stiffness. Sensors (Basel). 2023; 23: 9882. doi:10.3390/s23249882 https://doi.org/10.3390/s23249882 PMid:38139728 PMCid:PMC10747425 |
||||
| 6.Aimagambetova B, Ariko T, Merritt S, Rundek T. Arterial stiffness measured by pulse wave velocity correlated with cognitive decline in hypertensive individuals: A systematic review. BMC Neurol 2024; 24: 393. doi:10.1186/s12883-024-03905-8 https://doi.org/10.1186/s12883-024-03905-8 PMid:39415095 PMCid:PMC11481605 |
||||
| 7.Hellqvist H, Karlsson M, Spaak J, et al. Estimation of aortic stiffness by finger photoplethysmography using enhanced pulse wave analysis and machine learning. Front Cardiovasc Med 2024; 11: 1350726. doi:10.3389/fcvm.2024.1350726 https://doi.org/10.3389/fcvm.2024.1350726 PMid:38529332 PMCid:PMC10961400 |
||||
| 8.Xing X, Hong J, Alastruey J, et al. Robust arterial compliance estimation with Katz's fractal dimension of photoplethysmography. Front Physiol 2024; 15: 1398904. doi:10.3389/fphys.2024.1398904 https://doi.org/10.3389/fphys.2024.1398904 PMid:38915780 PMCid:PMC11194390 |
||||
| 9.Pilt K, Reiu A. Effect of transmural pressure on the estimation of arterial stiffness index from the photoplethysmographic waveform. Med Biol Eng Comput 2024; 62: 1049-59. doi:10.1007/s11517-024-02988-5 https://doi.org/10.1007/s11517-023-02992-y PMid:38123887 |
||||
| 10.Szołtysek-Bołdys I, Zielińska-Danch W, Łoboda D, Kowalski P, Nowak M, Wiśniewski R, et al. Do photoplethysmographic parameters of arterial stiffness differ depending on the presence of arterial hypertension and/or atherosclerosis? Sensors (Basel) 2024; 24: 4572. doi:10.3390/s24144572 https://doi.org/10.3390/s24144572 PMid:39065969 PMCid:PMC11280487 |
||||
| 11. Mazzolai L, Teixido-Tura G, Lanzi S, Boc V, Bossone E, Brodmann M, et al. ESC Scientific Document Group. 2024 ESC Guidelines for the management of peripheral arterial and aortic diseases. Eur Heart J 2024; 45: 3538-700. doi: 10.1093/eurheartj/ehae179 https://doi.org/10.1093/eurheartj/ehae179 PMid:39210722 |
||||
| 12.Hemla D, Agnoletti D, Jozwiak M, et al. Non-invasive estimation of central systolic blood pressure by radial tonometry: a simplified approach. J Pers Med 2023; 13: 1244. doi:10.3390/jpm13081244 https://doi.org/10.3390/jpm13081244 PMid:37623496 PMCid:PMC10455683 |
||||
Copyright

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
AUTHOR'S CORNER

