From atrium to ventricle - evolving evidence for extracardiac complications from non-thermal but not risk-free pulsed field ablation platform
EDITORIALS
From atrium to ventricle - evolving evidence for extracardiac complications from non-thermal but not risk-free pulsed field ablation platform
Article Summary
- DOI: 10.24969/hvt.2026.649
- CARDIOVASCULAR DISEASES
- Published: 05/05/2026
- Received: 01/05/2026
- Accepted: 01/05/2026
- Views: 27
- Downloads: 9
- Keywords: Pulse field ablation, complication, atrial fibrillation ablation, hemoptysis, hemolysis, extracardiac injury
Address for Correspondence: Narendra Kumar, HeartbeatsZ Academy, Norfolk, NR31 9DD, United Kingdom
Email: drnarendra007kr@gmail.com
ORCID: 0000-0002-4197-5133 Scopus Author ID: 55969944400
Editorial
From atrium to ventricle - evolving evidence for extracardiac complications from non-thermal but not risk-free pulsed field ablation platform
Narendra Kumar, HeartbeatsZ Academy, Norfolk, NR31 9DD, United Kingdom
Key words: Pulse field ablation, complication, atrial fibrillation ablation, hemoptysis, hemolysis, extracardiac injury
![]()

We read with interest the recent European Heart Journal – Case Reports publication by Mulder et al. (1) describing hemoptysis following atrial fibrillation (AF) ablation using pulse field ablation (PFA). The case is timely and clinically meaningful, drawing attention to a complication that, while uncommon, carries the potential for serious morbidity. Hemoptysis following AF ablation has classically been attributed to pulmonary vein (PV)-related injury, encompassing venous stenosis, bronchopulmonary vascular trauma, and inflammatory pulmonary hemorrhage. Our own group has previously documented hemoptysis arising days to weeks after cryoballoon ablation—a complication reported both in a peer-reviewed case report (2) and in subsequent communications in the Pakistan Journal of Cardiology (3, 4)—underscoring the capacity for subacute pulmonary injury to manifest in delayed fashion across thermal energy modalities. The present case extends this concern to PFA, reinforcing that the evolution toward non-thermal energy sources does not in itself eliminate the risk of extracardiac pulmonary injury.
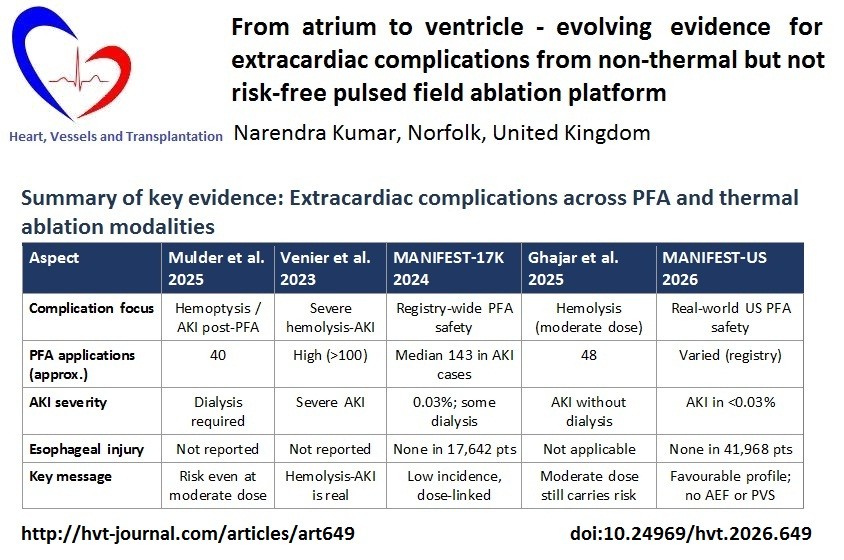
PFA achieves myocardial cell death through irreversible electroporation, delivering ultrashort high-voltage electrical pulses that selectively disrupt cardiomyocyte membranes while largely sparing adjacent structures such as the esophagus, phrenic nerve, and pulmonary vein walls (5). This tissue selectivity underpins the strong safety profile observed in large registries (Table 1). The MANIFEST-17K study, encompassing over 17,000 patients, reported no instances of atrio-esophageal fistula, pulmonary vein stenosis, or persistent phrenic nerve injury (6). The more recent MANIFEST-US study—the largest post-approval PFA dataset to date, including 41,968 patients from 102 United States centers—confirmed a major adverse event rate of only 0.63%, again with no cases of esophageal fistula, PV stenosis, or persistent phrenic paralysis (7). Taken together, these data confirm a favorable overall safety profile; nevertheless, they also reflect the limits of aggregate registry reporting when it comes to infrequent pulmonary complications.
Mild or self-limited hemoptysis, if attributed to anticoagulation alone or unreported, would not be captured. Early cross-sectional imaging—ideally contrast-enhanced computed tomography—remains essential in any post-ablation patient presenting with hemoptysis, irrespective of energy modality used.
Beyond pulmonary complications, PFA has been associated with a distinct pattern of systemic extracardiac injury: intravascular hemolysis with resultant hemoglobin-mediated nephrotoxicity and acute kidney injury (AKI). Severe hemolysis requiring dialysis has been described in individual case reports (8, 9) and was quantified in MANIFEST-17K at an incidence of 0.03% (5 of 17,642 patients). A recently published narrative review (March 2026) has clarified the mechanistic basis and procedural determinants of this complication, confirming that risk correlates with peak output voltage, the total number of energy applications delivered, and catheter-tissue contact quality (10). Critically, the threshold for clinically significant hemolysis appears to rise substantially when PFA applications exceed 90–100; patients with pre-existing renal impairment may reach this threshold at lower application counts. Separate work using tissue proximity indication software has further established that energy discharged from non-contact electrodes—irrespective of total pulse count—independently amplifies hemolysis markers, suggesting that catheter positioning discipline is as important as limiting the number of applications (11). The pathophysiological mechanisms underlying hemoptysis and hemolysis differ fundamentally; however, both complications serve as evidence that high-voltage electrical energy delivered within the left atrium can exert clinically meaningful effects at sites remote from the target myocardium.
Post-marketing surveillance data broaden the spectrum of extracardiac concern. A Medical Device Report submitted to the United States Food and Drug Administration documents a fatal atrio-esophageal fistula associated with PFA—a complication not captured in either MANIFEST registry—underscoring that rare but catastrophic outcomes may require longer follow-up and more granular reporting frameworks to surface reliably (12). A separate FDA MAUDE report describes a single episode of ventricular fibrillation occurring after the 11th pulsed field application during a PFA procedure, requiring emergency resuscitation. While individual adverse event reports cannot establish causality, they represent a critical adjunct to controlled registry data and should inform ongoing post-market surveillance protocols. Furthermore, gastroparesis has recently been identified as a potential extracardiac autonomic complication of pentaspline PFA even in the absence of endoscopically detectable esophageal injury, suggesting that vagal plexus modulation during posterior wall energy delivery may produce clinically relevant gastrointestinal effects not reflected in standard safety endpoints (13).
|
Table 1. Summary of key evidence: Extracardiac complications across PFA and thermal ablation modalities |
|||||
|---|---|---|---|---|---|
|
Aspect |
Mulder et al. 2025 |
Venier et al. 2023 |
MANIFEST-17K 2024 |
Ghajar et al. 2025 |
MANIFEST-US 2026 |
|
Complication focus |
Hemoptysis / AKI post-PFA |
Severe hemolysis-AKI |
Registry-wide PFA safety |
Hemolysis (moderate dose) |
Real-world US PFA safety |
|
PFA applications (approx.) |
40 |
High (>100) |
Median 143 in AKI cases |
48 |
Varied (registry) |
|
AKI severity |
Dialysis required |
Severe AKI |
0.03%; some dialysis |
AKI without dialysis |
AKI in <0.03% |
|
Esophageal injury |
Not reported |
Not reported |
None in 17,642 pts |
Not applicable |
None in 41,968 pts |
|
Key message |
Risk even at moderate dose |
Hemolysis-AKI is real |
Low incidence, dose-linked |
Moderate dose still carries risk |
Favourable profile; no AEF or PVS |
This observation expands the catalogue of PFA-specific extracardiac effects and warrants prospective study.
Contextualizing these findings within the broader catheter ablation literature is instructive.
A systematic review and meta-analysis of catheter ablation for valvular atrial fibrillation has demonstrated that procedural efficacy and safety outcomes differ meaningfully across structural substrates, with concomitant valvular pathology modifying both arrhythmia recurrence risk and the probability of procedural complications (5). This is directly relevant to PFA adoption, since real-world practice increasingly involves patients with structural heart disease, hypertensive cardiomyopathy, and impaired renal function—precisely the cohorts in whom hemolysis-related AKI may be most consequential and in whom post-ablation hemoptysis is least likely to be attributed to an ablation-related mechanism. The PFA-SHAM trial (7), presented at the American Heart Association 2025 Scientific Sessions, provided the first randomized placebo-controlled evidence that PFA reduces AF recurrence and improves quality of life compared with a sham procedure, with no major safety signals observed—strengthening the efficacy case while reinforcing that ongoing complication surveillance remains warranted as indications broaden.
The present report by Mulder et al. (1), read alongside our prior experience with cryoablation-related hemoptysis, and the emerging data on PFA-specific systemic complications, point to several practical conclusions. First, post-ablation hemoptysis must not be reflexively attributed to anticoagulation; early contrast-enhanced CT of the chest is essential regardless of energy source. Second, the risk of hemolysis-induced AKI should be anticipated prospectively in patients undergoing high-application-count PFA, particularly those with reduced baseline renal reserve, and procedural protocols should aim to minimize non-contact electrode energy delivery. Third, uncommon complications—whether pulmonary, renal, autonomic, or cardiac—are unlikely to be fully characterized within the observational follow-up windows of existing registries, and dedicated adverse event reporting, inclusive of delayed presentations, should be embedded within all post-approval PFA programs. As PFA platforms proliferate and indications expand, a rigorous and systematic approach to extracardiac safety remains indispensable to ensuring that the real-world experience matches the promise of pre-clinical and early registry data.
The emerging application of PFA to ventricular tachycardia (VT) ablation introduces a distinct and amplified complication landscape that warrants dedicated consideration. Unlike the thin-walled atrium, ventricular targets—particularly scar-related substrates—require deeper lesion penetration into fibrotic, heterogeneous myocardium, necessitating higher energy densities and greater application counts. This elevates the risk of hemolysis-induced AKI well beyond what is observed in AF ablation, while simultaneously raising concerns about coronary artery proximity, conduction system injury (including bundle branch block and atrioventricular block), and acute hemodynamic decompensation in patients who already carry severely impaired ventricular function.
A systematic review of PFA for VT identified vascular complications, cardiogenic shock, and conduction system damage as the principal risks in early-experience cohorts (14). The first-in-human VCAS trial, evaluating high-voltage focal PFA for scar-related VT, reported primary safety events in 11.5% of patients within 180 days—including cardiogenic shock and heart failure hospitalization—though no energy-related strokes or phrenic nerve injuries were observed, and hemolysis, while biochemically detectable, did not necessitate renal replacement therapy in any patient (15).
As VT PFA programs scale, prospective complication surveillance strategies must be specifically designed for the ventricular context, recognizing that the safety benchmarks established in AF cannot be directly extrapolated.
It will be worth monitoring once PFA will be used even for supraventricular tachycardia arrhythmias too.
Peer-review: Internal
Conflict of interest: None to declare
Authorship: N.K.
Acknowledgements and Funding: None to declare.
Statement on A.I.-assisted technologies use: Author stated they did not use artificial intelligence (A.I.) tools for preparation of the manuscript
Data and material availability: Do not apply
References
| 1. Mulder BA, Eisenga MF, Eijgelsheim M, Blaauw Y. Unexpected acute kidney injury requiring dialysis after routine pulsed field pulmonary vein isolation: A case report. Eur Heart J Case Rep 2025; ytaf669. https://doi.org/10.1093/ehjcr/ytaf669 PMid:41561780 PMCid:PMC12813576 |
||||
| 2. Kumar N, Ranganathan MK, Mustafa S, Saraf K, Timmermans C, Gupta D. Hemoptysis after cryoablation for atrial fibrillation. JAFIB 2019; 12: 2237. doi: 10.4022/jafib.1938 https://doi.org/10.4022/jafib.2237 PMid:32435347 PMCid:PMC7237077 |
||||
| 3. Kumar N. Complications following catheter ablation for atrial fibrillation: evolving perspectives. Pak J Cardiol 2025. | ||||
| 4. Kumar N. Catheter ablation of valvular atrial fibrillation: a systematic review and meta-analysis. Pak J Cardiol 2025. | ||||
| 5. Yabasigawa Y, Ibrahim W, Kumar N. A case of atrial fibrillation complicated by complete atrioventricular block. SAGE Open Med Case Rep. 2023. DOI: 10.1177/2050313X231157486 https://doi.org/10.1177/2050313X231157486 PMid:36866023 PMCid:PMC9972045 |
||||
| 6. Ekanem E, Reddy VY, Schmidt B, Kautzner J, van der Voort P, Jais P, et al. Safety of pulsed field ablation in more than 17,000 patients with atrial fibrillation in the MANIFEST-17K study. Nat Med 2024; 30: 2020-9. doi: 10.1038/s41591-024-03114-3 https://doi.org/10.1038/s41591-024-03114-3 PMid:38977913 PMCid:PMC11271404 |
||||
| 7. Turagam MK, Aryana A, Day JD, Dukkipati SR, Hounshell T, Nair D, et al. Multicenter Study on the Safety of Pulsed Field Ablation in Over 40,000 Patients: MANIFEST-US. J Am Coll Cardiol 2026; 87: 172-93. DOI: 10.1016/j.jacc.2025.10.051 https://doi.org/10.1016/j.jacc.2025.10.051 PMid:41389071 |
||||
| 8. Venier S, Vaxelaire N, Jacon P, Carabelli A, Desbiolles A, Garban F, et al. Severe acute kidney injury related to haemolysis after pulsed field ablation for atrial fibrillation. Europace 2023; 26: euad371. DOI: 10.1093/europace/euad371 https://doi.org/10.1093/europace/euad371 PMid:38175788 PMCid:PMC10776308 |
||||
| 9. Ghajar A, Uzunoglu EC, Lopes J, Assis F, Nekkanti R, Popescu SS, et al. Severe hemolysis after moderate-dose pulsed field application for atrial fibrillation/flutter: a cautionary tale. JACC Case Rep 2025; 30: 105136. doi: 10.1016/j.jaccas.2025.105136 https://doi.org/10.1016/j.jaccas.2025.105136 PMid:40940117 PMCid:PMC12789745 |
||||
| 10. Zhou M, Yang H, Sun H, Yu M, Si D, He Y. Hemolysis in pulsed field ablation for atrial fibrillation: A narrative review. Rev Cardiovasc Med 2026; 27: doi: 10.31083/rcm46485 https://doi.org/10.31083/RCM46485 PMid:41923751 PMCid:PMC13036529 |
||||
| 11. Yoshimura S, Kaseno K, Kodama A, Nishiuci S, Hattori K, Masuyama T, et al. Effect of pulsed field ablation delivered from noncontact catheter electrodes on hemolysis: A tissue proximity indication-based analysis. Heart Rhythm O2 2025; 6: 1716-21. doi: 10.1016/j.hroo.2025.09.002 https://doi.org/10.1016/j.hroo.2025.09.002 PMid:41357276 PMCid:PMC12675092 |
||||
| 12. U.S. Food and Drug Administration. MAUDE Adverse Event Report: Medtronic, Inc. AFFERA; Percutaneous Cardiac Ablation Catheter for Treatment of Atrial Fibrillation. MDR Report Key 23733351. Published 24 December 2025. | ||||
| 13. Haj Abdo M, Deneke T, Nentwich K, Chakarov I, Berkovitz A, Sauer E, et al. Gastroparesis as a potential complication of pentaspline pulsed field ablation without endoscopic esophageal injury. J Interv Card Electrophysiol 2026; 69: 63-9. doi: 10.1007/s10840-025-02130-8 https://doi.org/10.1007/s10840-025-02130-8 PMid:40974470 |
||||
| 14. Askarinejad A, Kohansal E, Sabahizadeh A, Hesami H, Adimi S, Haghjoo M. Pulsed-field ablation in management of ventricular tachycardia: a systematic review of case reports and clinical outcomes. Clin Cardiol 2024; 47: e70018. doi: 10.1002/clc.70018 https://doi.org/10.1002/clc.70018 PMid:39350646 PMCid:PMC11442986 |
||||
| 15. Reddy VY, Koruth JS, Peichl P, Petru J, Funasako M, Skoda J, et al. High-voltage focal pulsed field ablation to treat scar-related ventricular tachycardia: the first-in-human VCAS trial. Circulation 2025; 152: 1691-704. doi: 10.1161/CIRCULATIONAHA.125.07702 https://doi.org/10.1161/CIRCULATIONAHA.125.077025 PMid:41071961 PMCid:PMC12700701 |
||||
Copyright

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
AUTHOR'S CORNER

