Comparison of the diagnostic utility of computed tomography venography with color Doppler ultrasound in patients suspected of secondary varicose veins: Study on diagnostic accuracy
ORIGINAL RESEARCH ARTICLE
Comparison of the diagnostic utility of computed tomography venography with color Doppler ultrasound in patients suspected of secondary varicose veins: Study on diagnostic accuracy
Article Summary
- DOI: 10.24969/hvt.2026.652
- CARDIOVASCULAR DISEASES
- Published: 17/05/2026
- Received: 13/04/2026
- Revised: 06/05/2026
- Accepted: 07/05/2026
- Views: 10
- Downloads: 4
- Keywords: Varicose veins, CT venography, color Doppler ultrasound, deep venous thrombosis, May-Thurner syndrome, pelvic congestion syndrome, iliac veins
Address for Correspondence: Mosab Ahmed Abdelsalam, Department of Diagnostic and Interventional Radiology, Faculty of Medicine, Qena University, Qena, Egypt
Email: Mos3aba44@gmail.com Phone: +20 01093424500
ORCID: 0000-0003-0635-2712 Facebook: @mosaab.ahmed.5
Abd-Alraheem Hussein Ali 1, Abd-Alraheem Fathy 2, Ahmed Okasha Mohammed 1, Mosab Ahmed Abdelsalam 1
1Department of Diagnostic and Interventional Radiology, Faculty of Medicine, Qena University, Qena, Egypt
2Department of Vascular Surgery, Faculty of Medicine, Qena University, Qena, Egypt
Abstract
Objective: This study aimed to evaluate the diagnostic utility of computed tomography venography (CTV) compared to Color Doppler Ultrasound (CDU) in patients with suspected secondary varicose veins.
Methods: This prospective diagnostic accuracy study included 100 consecutive patients with clinically suspected secondary lower limb varicosities evaluated between 2023 and 2025. Patients underwent detailed clinical examination, CDU, and 128-detector CTV. Evaluated parameters included the precise anatomical distribution of varicosities, central venous compression syndromes, and diagnostic agreement between the two modalities.
Results: The cohort comprised 51 (51%) males and 49 (49%) females (mean age: 36.4 (11.29) years). CTV definitively diagnosed May-Thurner syndrome in 31% of patients, pelvic congestion syndrome in 25%, lower limb deep venous thrombosis (DVT) in 12%, iliac venous thrombosis in 10%, Nutcracker syndrome in 5%, Klippel-Trenaunay syndrome in 4%, portal hypertension in 3%, Budd-Chiari syndrome, and inferior vena cava thrombosis in 2%. CDU failed to visualize the iliac veins in 60% of cases and was severely limited by patient obesity in 40%. For the detection of ovarian vein dilatation, CDU demonstrated a sensitivity of 93.3%, specificity of 80%, and an overall accuracy of 88% compared to CTV, with substantial agreement (κ = 0.73).
Conclusion: While CDU remains highly accurate for localized superficial and gonadal screening, it is insufficient for mapping central and peripheral venous pathologies. CTV is a method to be considered in patients with suspected secondary varicose veins, and in those whose causes cannot be determined by CDU.
Key words: Varicose veins, CT venography, color Doppler ultrasound, deep venous thrombosis, May-Thurner syndrome, pelvic congestion syndrome, iliac veins

Graphical abstract

Introduction
Traditional phlebography has gradually been replaced by more sophisticated non-invasive imaging modalities. One such technique that has shown promise is direct computed tomography venography (CTV). CTV enables accurate visualization and assessment of the precise location, size, and extent of venous lesions (1). A notable advantage of CTV over traditional Color Doppler Ultrasound (CDU) lies in its capability to effectively capture and detect venous abnormalities in both the pelvic and calf regions—areas that pose significant diagnostic challenges for CDU alone (2).
Current 2025-2026 guidelines from the Society for Cardiovascular Angiography and Intervention and the Society of Interventional Radiology recommendation are: CDU is the cornerstone for chronic venous disease (CVD); essential for assessing venous flow dynamics, locating reflux, and identifying anatomical abnormalities, transabdominal and transvaginal CDU is the preferred first-line imaging for pelvic venous disorders due to its ability to dynamically assess venous flow, reflux, and visualize periuterine/periovarian varices (3).
CTV plays a crucial role in assessing varicose veins, a condition affecting 10%–40% of individuals aged 30–70 years. Determining the underlying cause of venous insufficiency is critical. In cases where varicose veins exhibit complex morphology or unusual manifestations, combining CDU with CTV aids in understanding the exact characteristics and causes of varicosities (4). By employing CTV, clinicians can acquire high-resolution images that reveal precise anatomical information (5), enabling a comprehensive evaluation of the vascular lesions and guiding targeted therapeutic strategies (6). While the diagnosis of deep vein thrombosis (DVT) and evaluation of iliac vein patency have historically been performed with CDU, helical CTV yields objective information about underlying anatomic abnormalities crucial for accurate diagnosis and evaluation of post-interventional thrombosis (7–9).
This study aimed to evaluate the diagnostic utility of CTV compared to CDU in patients with suspected secondary varicose veins.
Methods
Study design and population
Study design and setting: This prospective diagnostic accuracy study was conducted on 100 patients admitted to Qena University Hospitals between 2023 and 2025.
Participants: The inclusion criteria comprised patients of all ages and both sexes with clinically suspected secondary varicose veins (e.g., pelvic congestion syndrome, DVT, Klippel-Trenaunay syndrome, May-Thurner syndrome). Morbidly obese patients in whom standard CDU were technically inadequate were also included. Patients with primary varicose veins, neoplastic/traumatic venous disease, prior venous surgery, or contraindications to CT contrast (severe renal impairment, pregnancy, allergy) were excluded. Patients were consecutively enrolled during the study period to reduce selection bias and better reflect real-world clinical practice.
Ethics: The study was approved by the Institutional Ethics Committee. Written informed consent was obtained from all participants. The study was conducted in compliance with Helsinki 2024 agreement.
Baseline variables
Detailed medical histories and clinical examinations were conducted. Baseline data included: age, body mass index (BMI), gender, parity (in female), lower limb varicose veins, pain, and edema, varicocele, vulvular varices, infertility, telangiectasia, hematuria, limb discrepancy, venous ulcers, and biles. Laboratory investigations included coagulation profiles, D-dimer, and renal function tests (serum creatinine via Jaffe’s Kinetic method).
Clinical examinations for varicose veins
Complaints as pain and edema were recorded. Medical history and physical examination signs for lower limb varicose veins, pain, and edema, varicocele, vulvular varices were obtained. Examinations were performed in standing and supine positions to map varicosities, pigmentation, and ulceration.
Imaging protocols for varicose veins
CT Venography
CTV was performed using a GE 128-detector row CT scanner (GE Healthcare, Chicago, IL, USA), (120 kV, 215 mA, pitch ≤ 1, 0.5 s rotation time, 0.6 mm slice thickness). Non-ionic iodinated contrast agent (iopromide 300 mg iodine/mL; Ultravist ® 300, Bayer Healthcare, Berlin, Germany) was injected at 4–5 mL/s (maximum 100 mL for adults) followed by a 50–60 mL saline flush. Bolus-tracking initiated scanning when femoral vein enhancement reached 120 HU. Scans extended from the diaphragm to the feet.
The evaluated CTV variables included venous compression syndromes (e.g., May-Thurner syndrome and Nutcracker syndrome), DVT, iliac and inferior vena cava (IVC) patency, pelvic venous congestion, ovarian vein dilatation, abdominal and pelvic collateral pathways, perforator vein mapping, venous reflux-related morphological changes, and associated anatomical variants or extrinsic compressive causes.
Color Doppler ultrasonography
CDU was conducted using a high-resolution ultrasound system (LOGIQ P7, GE Healthcare, Chicago, IL, USA) equipped with a 5-10 MHz linear transducer probe. Superficial, deep, and perforator veins were systematically assessed in standing (reflux) and supine (thrombosis) positions. Reflux was defined as retrograde flow >0.5 seconds.
CDU assessment included superficial and deep venous patency, lower limb deep vein thrombosis, saphenofemoral junction reflux, perforator incompetence, ovarian vein dilatation when visualized, pelvic venous congestion, and venous flow abnormalities.
Hemodynamically significant venous compression was defined as ≥50% reduction in luminar diameter compared to the adjacent normal venous segment.
Statistical analysis
Data were collected, coded, revised, and entered into the Statistical Package for the Social Sciences (SPSS) software program, version 27 (SPSS Inc., Chicago, IL, 2021). Qualitative variables are presented as numbers and percentages and compared using a McNemar test. Quantitative variables were tested for normality using the Kolmogorov–Smirnov and Shapiro–Wilk tests. Normally distributed numerical variables are reported as mean (SD) and range. Diagnostic performance (sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), and accuracy) was assessed. Cohen’s Kappa was used for the agreement between CDU and CTV. Significance was set at p < 0.05.
Compliance with STROBE and STARD guidelines
The study was prepared in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines and the Standards for Reporting Diagnostic Accuracy Studies (STARD) recommendations.
Results
Baseline demographic and clinical characteristics
This prospective study included 100 patients evaluated for secondary lower limb varicose veins. Table 1 represents the demographics and clinical signs.
|
Table 1. Basic demographic data and clinical characteristics of the patient population (n =100) |
||
|
*Age, years |
36.4 (11.29) |
|
|
Gender, n(%) |
Male |
51 (51) |
|
Female |
49 (49) |
|
|
*BMI, kg/m2 |
25.7 (4.53) |
|
|
Parity (49 female), n(%) |
Nulliparous |
8 (16.3) |
|
Multiparous |
41 (83.7) |
|
|
Lower limb varicose veins, n(%) |
97 (97) |
|
|
Lower limb pain, n(%) |
36 (36) |
|
|
Varicocele (51 male) , n(%) |
26 (50.98) |
|
|
Lower limb edema and redness , n(%) |
9 (9) |
|
|
Vulvular varices (49 female) , n(%) |
7 (14.29) |
|
|
Infertility, n(%) |
8 (8) |
|
|
Venous ulcer, n(%) |
4 (4) |
|
|
Telangiectasia, n(%) |
7 (7) |
|
|
Hematuria, n(%) |
7 (7) |
|
|
Hematemesis, n(%) |
7 (7) |
|
|
Menorrhagia, n(%) |
3 (3) |
|
|
Limb discrepancy, n(%) |
4 (4) |
|
|
Biles, n(%) |
7 (7) |
|
|
Known liver cirrhosis, n(%) |
7 (7) |
|
|
Data are presented as *mean (SD) and number (%) BMI- body mass index |
||
The cohort was evenly distributed by sex, comprising 51 (51%) males and 49 (49%) females. The mean age was 36.4 (11.29) years (range: 6–60 years), with a mean BMI of 25.7 (4.53) kg/m² (range: 18–40.6 kg/m²). Among the female participants, 41 (83.7%) were multiparous and 8 (16.3%) were nulliparous.
Clinically, lower limb varicosities were the predominant physical sign, present in 97% of the cohort. Gender-specific clinical manifestations included male varicocele, identified in 50.9% of male patients (26/51), and vulvar varices, observed in 7% of the total cohort.
A history of infertility was reported in 8% of patients, including both male and female individuals, predominantly female participants, due to the relation to underlying pelvic venous pathology. Symptomatically, patients presented with lower limb pain (36%), localized edema and redness (9%), telangiectasia (7%), and advanced venous ulcers (4%). Systemic and atypical manifestations were also recorded, including hematuria (7%), hematemesis (7%), biliary involvement (7%), known liver cirrhosis (7%), limb discrepancy (4%), and menorrhagia (3%).
CTV findings
Left common iliac vein compression (Fig. 1) was observed in 43% of patients, with the degree of stenosis ranging from 15% to 90% (mean: 55.1 (21.08)%).
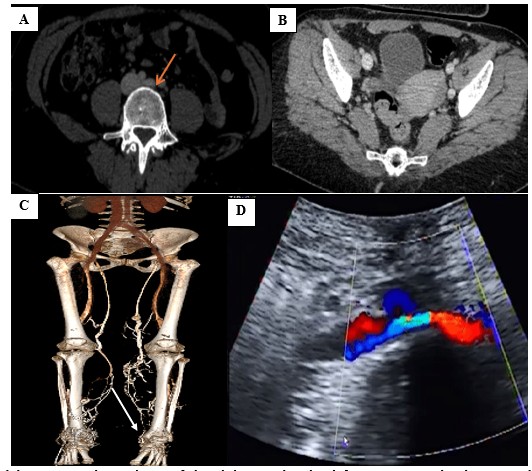
Figure 1. (A) Axial contrast enhanced CTV of the abdomen showing left CIV compression between right CIA and the lumbar vertebral body, with luminal narrowing of the left CIV (orange arrow); (B) Axial contrast enhanced CTV showing dilated tortious congested pelvic veins; (C) 3D reconstruction showing the compressed left CIV and bilateral lower limb varicosities. (D) CDU showing compression of the left CIV (white arrow). These findings are consistent with May-Thurner syndrome (non-thrombotic type)
CDU - color Doppler ultrasound, CIA - common iliac artery, CIV - common iliac vein, CTV – computed tomography venography
Overall venous compression within the cohort was classified as hemodynamically significant in 76% of cases, borderline in 1%, and non-significant in 23%. Renal vein compression observed in 4% of patients was consistent with Nutcracker syndrome, characterized by compression of the left renal vein between the superior mesenteric artery and the abdominal aorta. Deep venous reflux was highly prevalent, identified in 65% of the cohort.
It was inferred based on indirect imaging features, including venous dilatation, collateral pathways, and associated pelvic congestion patterns.
Pelvic varices and congestion were detected in 30% of patients, while abdominal and pelvic wall varicosities were noted in 34%. Among female participants, substantial ovarian vein dilatation was present in 61.2% (30/49). Thrombotic events were accurately mapped, with lower limb DVT present in 20% of patients and IVC thrombosis in 2%.
Detailed mapping of perforator veins demonstrated that leg and calf perforators were nearly ubiquitous (95%). Pelvic and gluteal perforators were identified in 15% of patients, followed by obturator (16%), inguinal (7%), lumbar (5%), lateral vein of Servelle (4%), sacral (3%), and pudendal (3%) perforators. Rare collateral pathways, including intrahepatic and azygos collaterals, were observed in 1% each.
Secondary anatomical variants and extrinsic factors identified via CTV included inguinal lymphadenopathy (9%), a bulky uterus (8%), uterine fibroids (12.2%), duplicated IVC (4%), left-sided IVC (4%), and retro-aortic renal vein (3%). One patient (1%) presented with an IVC JJ stent migration.
Final anatomical diagnoses
Based on comprehensive CTV findings, May-Thurner syndrome was the leading definitive diagnosis, present in 31% of the cohort (Table 2). This was followed by pelvic congestion syndrome (25%) (Fig. 2), lower limb venous thrombosis (12%), and iliac venous thrombosis (10%). Other diagnoses included Nutcracker syndrome (5%), primary varicose veins reclassified after ruling out secondary causes (6%), Klippel-Trenaunay syndrome (4%), portal hypertension (3%), IVC thrombosis (2%), and Budd-Chiari syndrome (2%).
|
Table 2. Main final diagnoses on CTV of the studied patients (n =100) |
|
|
Variables |
n(%) |
|
May-Thurner syndrome |
31 (31) |
|
Pelvic congestion syndrome |
25 (25) |
|
Iliac venous thrombosis |
10 (10) |
|
IVC thrombosis |
2 (2) |
|
Lower limb venous thrombosis |
12 (12) |
|
Nutcracker syndrome |
5 (5) |
|
Primary varicose veins (no secondary cause) |
6 (6) |
|
Portal hypertension |
3 (3) |
|
Klippel- Traunany syndrome |
4 (4) |
|
Budd-Chiari syndrome |
2 (2) |
|
Data are presented as number (%) CTV – computed tomography venography, IVC – inferior vena cava |
|
Incidental extravascular findings were common and included splenomegaly (5%), ovarian cysts (5%), hydronephrosis (4%), gallbladder stones (4%), incidental liver cirrhosis (4%), paraesophageal varices (4%), ureteric stones (3%), left ovarian cystadenoma (3%), portal hypertension (2%), and enlarged congested prostate (2%). Prior trauma-related findings included right above-knee amputation (3%) and right iliac bone fracture via gunshot (2%).
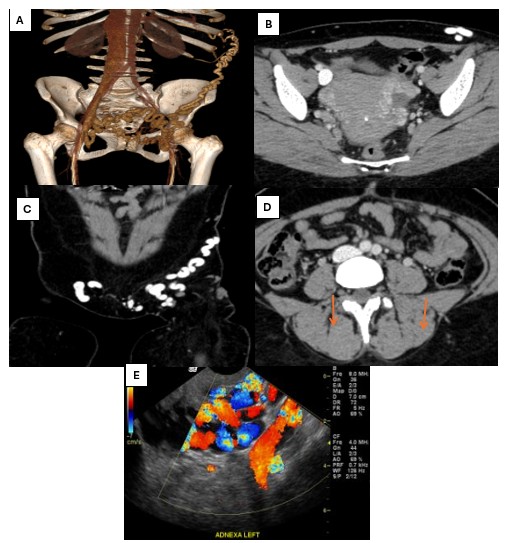
Figure 2. (A) 3D reconstruction of CTV showing multiple dilated tortious superficial and deep pelvic veins; (B) Axial CTV showing multiple dilated enhanced parametrial and para-ovarian pelvic plexus; (C) Coronal contrast enhanced CTV of the pelvis showing dilated tortious left anterior superficial pelvic veins; (D) Note the dilated ovarian veins anterior to the psoas muscles on both sides (orange arrows).; (E) CDU showing dilated left adnexal veins with reflux during Valsalva maneuver. Findings are consistent with pelvic congestion syndrome.
CDU – color Doppler ultrasound, CTV – computed tomography venography
CDU findings
CDU successfully identified lower limb varicosities in 98% of patients. However, its detection of complex pathology was notably lower than CTV, observed as right iliac and deep pelvic thrombosis as shown in (Fig. 3), leg perforators were identified in 33%, saphenofemoral junction (SFJ) reflux in 32%, lower limb DVT in 9%, and compressed left common iliac vein in 18%. Central pelvic findings via CDU included deep pelvic venous congestion (14%), iliac/deep pelvic thrombosis (10%), and ovarian vein dilatation (9%) (Table 3).
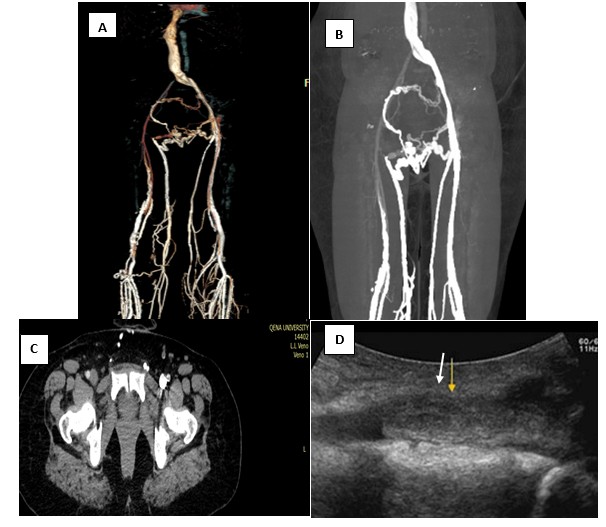
Figure 3. (A) 3D reconstruction of CTV showing absent right CIV, IIV and EIV with multiple superficial and deep pelvic venous tortious collaterals; (B) Coronal CTV of the pelvic and both lower limbs veins; (C) Contrast enhanced CTV of the pelvis showing filling defect within the right EIV (white arrow) (note the difference of the contrast opacification of the normal left EIV). (D) CDU showing left iliac DVT (yellow arrow). Findings consistent with extensive right deep pelvic venous thrombosis.
CDU – color Doppler ultrasound, CIV-common iliac vein, CTV – computed tomography venography, EIV- external iliac vein, IIV - internal iliac vein
Comparative diagnostic performance
When evaluating ovarian vein dilatation and pathological reflux (among 49 female patients) using CTV as the reference standard, CDU correctly identified 27 true positive and 16 true negative cases. This yielded a sensitivity of 93.3%, specificity of 80.0%, PPV of 87.5%, and NPV of 88.9%, with an overall accuracy of 88.0%. Agreement between the two modalities for this specific metric was substantial (κ = 0.73) (Table 4).
For the prediction of the final overall diagnosis, CDU correctly identified 82 true positives and 12 true negatives against the CTV gold standard. CDU demonstrated a sensitivity of 97.6%, specificity of 75.0%, PPV of 95.3%, and NPV of 85.7%, with an overall diagnostic accuracy of 94.0% (p < 0.001; McNemar test). The inter-modality agreement was substantial (κ = 0.74) (Table 5).
|
Table 3. Color Doppler ultrasonography findings of the studied patients (n = 100) |
|
|
Variables |
n(%) |
|
Lower limb varicosities |
98 (98) |
|
Lower limb DVT |
9 (9) |
|
Leg Perforators |
33 (33) |
|
SFJ reflux |
32 (32) |
|
Ovarian vein dilatation |
9 (9) |
|
Deep pelvic venous congestion |
14 (14) |
|
Iliac and deep pelvic thrombosis |
10 (10) |
|
Compressed left CIV |
18 (18) |
|
Absent flow in IVC and hepatic veins |
4 (4) |
|
Dilated PV |
3(3) |
|
Compressed left renal vein |
4 (4) |
|
Liver cirrhosis |
7 (7) |
|
Arterial abdominal and pelvic wall varices |
14 (14) |
CIA- Common iliac artery, CIV- Common iliac vein, DVT- deep vein thrombosis, PV- Portal vein, SFJ- saphenofemoral junction |
|
|
Table 4. Comparison of diagnostic performance of CDU and CT venography for ovarian vein dilation and pathological reflux (49 female patients) |
||||
|
CDU ovarian vein findings |
CTV |
Total |
Diagnostic performance |
|
|
Positive |
Negative |
|||
|
Positive |
27 (TP) |
3 (FP) |
30 |
Sensitivity: 93.3% |
|
Negative |
3 (FN) |
16 (TN) |
19 |
Specificity: 80.0% |
|
Total |
30 |
19 |
49 |
|
|
PPV: 87.5% |
NPV: 88.9% |
Accuracy: 88.0% |
Kappa (κ): 0.73 |
|
|
CDU – color Doppler ultrasound, CTV – computed tomography venography, FN – false-negative, FP – false-positive, NPV – negative predictive value, PPV – positive predictive value, TN – true negative, TP – true positive |
||||
|
Table 5. Comparison of CDU and CTV in prediction of final diagnosis |
||||
|
CDU |
CTV |
p |
||
|
Positive, n(%) |
Negative, n(%) |
Total |
||
|
Positive, n(%) |
82(82%) |
4(4%) |
86 |
<0.001* |
|
Negative, n(%) |
2(2%) |
12(12%) |
14 |
|
|
Total |
84 |
16 |
100 |
|
|
Specificity: 75% Sensitivity: 97.6% |
PPV: 95.3% |
NPV: 85.7% |
Accuracy: 94% |
Kappa: 0.74 |
|
Data are presented as numbers and number (%), *McNemar test CDU – color Doppler ultrasound, CTV – computed tomography venography, NPV – negative predictive value, PPV – positive predictive value |
||||
Discussion
This prospective study was conducted on 100 patients presenting with clinical suspicion of secondary lower limb varicose veins from Qena University Hospital between 2023 and 2025, and evaluated the diagnostic utility of CTV compared to CDU in patients with suspected secondary varicose veins. Among the 100 studied patients, 51% were males and the mean age was 36.4 (11.29) years. Among the 39 female patients, 83.7% were multiparous.
Our study demonstrated CDU has a good agreement with CTV findings with diagnostic accuracy in identifying secondary varicose veins of 94%. CDU however is highly accurate for superficial vein and gonadal varices, while low value in identification of deep venous structures. CTV offers precise visualization of deep anatomical structures, diagnosis of lower limb varicose veins, pelvic syndromes and central obstructive pathologies.
The anatomy of the lower limb venous system is highly complex, making the assessment of venous insufficiency a diagnostic challenge. Varicose veins affect 10%–40% of the adult population and are rooted in venous valvular dysfunction (10). While CDU offers dynamic, real-time assessment (11), CTV adds significant value by identifying extrinsic compressions and complex congenital malformations often missed by ultrasound (4).
The cohort's mean BMI (25.7 kg/m²) and high multiparity rate reflect the cumulative effects of venous hypertension and structural vessel wall degradation. Obesity raises intra-abdominal pressure, and multiple pregnancies mechanically compress iliac veins, promoting permanent venous dilation (12). The slight male predominance (51%) aligns with occupational findings by Elumalai et al. (13) contrasting with the general clinical female predominance reported by Mulita et al. (14). The association between higher BMI and varicosities in the present study is strongly supported by Mok Ballew et al. (15) and Fusco et al., 2026 (16), reinforcing the heterogeneity of venous pathology across patient populations.
Clinically, the high incidence of male varicocele (50.9%), vulvar varices, and infertility points to upstream pathology like pelvic congestion syndrome or Nutcracker syndrome. Furthermore, portal hypertension manifestations (hematemesis, cirrhosis) increase systemic venous load (17). Doğan et al. (18) reported varicocele in 34.3% of patients with saphenous reflux, supporting elevated findings among study participants. Ulcer prevalence (4%) closely mirrored the 4.8% reported by Mulita et al. (14), though edema rates were lower in the current study. Clark and Taylor (19) confirm pelvic venous disorders affect approximately 15% of women, consistent with demographics in the current study.
Anatomically, left common iliac vein compression (43%) and reflux (65%) dominated the findings, driving a pathophysiology rooted in proximal venous outflow obstruction (20). Gavrilov (21) found pelvic congestion syndrome in 75.5% of women with pelvic varices, reinforcing the high pelvic disease burden observed in the current study. Wang et al. (22) noted left common iliac vein compression in 55.7% of patients, closely matching the 43% rate in the present study. Incompetent perforators were widespread (95%), a rate higher than the 58% reported by Zhang et al. (23), likely reflecting the combined imaging approach's higher sensitivity in this study.
Incidental findings such as splenomegaly and liver cirrhosis highlight systemic mechanisms elevating venous pressure (24).
The core diagnostic disparity lies in reflux detection: CTV identified reflux in 65% (deep/pelvic), while CDU found it in only 32% (predominantly superficial) (25). The prevalence of May-Thurner syndrome (31%) exactly matches findings by Heller et al. (26). Osman et al. (27) confirmed CT's high sensitivity (94.8%) for pelvic congestion syndrome. Conversely, baseline compression in unselected patients is much lower (14.7%), per Corrêa et al. (28). Badawy et al. (29) validate the heavy burden of iliac compression in symptomatic cohorts.
CDU failed to identify iliac veins in 60% of patients. This is driven by acoustic attenuation from deep adipose tissue and acoustic shadowing from bowel gas (30). Literature corroborates that CDU often underestimates iliac stenosis with high false-positive rates compared to cross-sectional modalities (31). In contrast, Clode et al. (32) found CDU sufficient for iliac stent evaluation in 99% of their cohort despite obesity, highlighting a divergence depending on the specific post-operative vs. diagnostic setting. We emphasize that CTV may be most appropriately utilized in selected patients, particularly in cases where CDU findings are inconclusive or when more detailed anatomical evaluation is required.
Strengths and limitations of the study
The primary strength is the prospective, dual-modality design. Limitations include the single-center setting and the inherent risks of ionizing radiation and contrast administration associated with CTV.
The diagnostic utility of CDU was substantially limited by patient habitus and deep anatomical structures. Obesity limited the sonographic window in 40% of cases. Consequently, CDU failed to identify the internal iliac veins in 60% of patients, the renal veins in 30%, and the ovarian veins in 32.65% of female patients.
Conclusion
Computed tomography venography provides indispensable anatomical mapping in the evaluation of secondary lower limb varicose veins, definitively identifying central obstructive pathologies such as May-Thurner and pelvic congestion syndromes. While Color Doppler Ultrasonography remains highly accurate for localized superficial and gonadal screening, its high failure rate in visualizing deep pelvic structures limits its standalone utility. A larger-scale, multicenter randomized trials are recommended for more informative findings.
Ethics: Written informed consent was obtained from all participants. This study was approved by the Institutional Ethics Committee of Qena University Hospitals and conducted in accordance with the Declaration of Helsinki (2024).
Peer-review: External and internal
Conflict of interest: None to declare
Authorship: Study concept and design: all authors; Data acquisition: A.-A.H.A., A.O.M., Data analysis: M.A.A., manuscript drafting: M.A.A.; Critical revision and approval final version for publication: all authors. Thus all authors fulfilled authorship criteria.
Acknowledgements and Funding: None to declare.
Statement on A.I.-assisted technologies use: No artificial intelligence tools were used for image generation, data analysis, or scientific decision-making. Any language assistance tools used during manuscript preparation did not influence the scientific content of the study.
Data and material availability: De-identified participant data supporting the findings of this study are available from the corresponding author upon reasonable academic request. Any sharing should in frame of fair use an collaboration with acknowledgement of source
References
| 1.Lan YQ, Xi ZF, Dong JJ, Chen YM, Wang YF, Feng N. Added value of computed tomography venography in the identification of abnormities in veins of lower extremities. Curr Med Res Opin 2022; 38: 927-36. doi:10.1080/03007995.2022.2057151 https://doi.org/10.1080/03007995.2022.2057151 PMid:35321594 |
||||
| 2. Mavili E, Ozturk M, Akcali Y, Donmez H, Yikilmaz A, Tokmak TT, et al. Direct CT venography for evaluation of the lower extremity venous anomalies of Klippel-Trenaunay Syndrome. AJR Am J Roentgenol XXXX; 192: XX-X. doi:10.2214/AJR.08.1151 https://doi.org/10.2214/AJR.08.1151 PMid:19457795 |
||||
| 3.Kaufman C, Winokur RS, Khilnani N, Pereira K, Taylor AC, Gupta R, et al. The society of interventional radiology practice guidance document on venous origin chronic pelvic pain in women. J Vasc Interv Radiol 2026; 107954. doi:10.1016/j.jvir.2025.107954 https://doi.org/10.1016/j.jvir.2025.107954 PMid:41786126 |
||||
| 4. Shi WY, Wang LW, Wang SJ, Yin XD, Gu JP. Combined direct and indirect CT Venography (Combined CTV) in detecting lower extremity deep vein thrombosis. Medicine 2016; 95: e3010. doi:10.1097/MD.0000000000003010 https://doi.org/10.1097/MD.0000000000003010 PMid:26986113 PMCid:PMC4839894 |
||||
| 5.Varghese R, Patel M, Rajarshi M, Khanna AK. Varicose veins-How to investigate. Indian J Surg 2021; 85: 15-21. doi:10.1007/S12262-021-03093-6 https://doi.org/10.1007/s12262-021-03093-6 |
||||
| 6.Adler C, Mousa A, Rhee A, Patel MD. Varicose veins of the lower extremity: doppler us evaluation protocols, patterns, and pitfalls. Radiographics 2022; 42: 2184-200. doi:10.1148/RG.220057/RG220057SUPPM5.MP4 https://doi.org/10.1148/rg.220057 PMid:36178804 |
||||
| 7.Finkelstein ER, Crist TE, Shao T, Mella-Catinchi J, Xu KY. The utility of computed tomography venography in the routine evaluation of patients who present to a lymphedema center with lower extremity edema. J Vasc Surg Venous Lymphat Disord 2023; 11: 1055-62. doi:10.1016/j.jvsv.2023.05.008 https://doi.org/10.1016/j.jvsv.2023.05.008 PMid:37196921 |
||||
| 8.O'Sullivan GJ, Semba CP, Bittner CA, Stephen TK, Razavi MK, Sze DY, et al. Endovascular management of iliac vein compression (May-Thurner) syndrome. J Vasc Interv Radiol 2000; 11: 823-36. doi:10.1016/S1051-0443(07)61796-5 https://doi.org/10.1016/S1051-0443(07)61796-5 PMid:10928517 |
||||
| 9.Jeon UB, Chung JW, Jae HJ, Kim HC, Kim SJ, Ha J, et al. May-Thurner syndrome complicated by acute iliofemoral vein thrombosis: helical CT venography for evaluation of long-term stent patency and changes in the iliac vein. AJR Am J Roentgenol 2010; 195: 751-7. doi:10.2214/AJR.09.2793 https://doi.org/10.2214/AJR.09.2793 PMid:20729456 |
||||
| 11.Knuttinen MG, Naidu S, Oklu R, Kriegshauser S, Eversman W, Rotellini L, et al. May-Thurner: diagnosis and endovascular management. Cardiovasc Diagn Ther 2017; 7(Suppl 3): S159. doi:10.21037/CDT.2017.10.14 https://doi.org/10.21037/cdt.2017.10.14 PMid:29399519 PMCid:PMC5778514 |
||||
| 12.Raffetto JD. Pathophysiology of chronic venous disease and venous ulcers. Surg Clin North America 2018 ; 98: 337-47. doi:10.1016/j.suc.2017.11.002 https://doi.org/10.1016/j.suc.2017.11.002 PMid:29502775 |
||||
| 13.Elumalai V, Ningombam JD, Sahu M, Banerjee S. Prevalence and risk factors of varicose veins in a tertiary care hospital in Pondicherry: A cross-sectional study. Indian J Public Health Res Dev 2025; 16: 290-6. doi:10.37506/AS57QF83 https://doi.org/10.37506/as57qf83 |
||||
| 14.Mulita F, Dimopoulos P, Verras GI, Mustaqe P, Jaho J, Leivaditis V, et al. Demographic and clinical characteristics of patients with varicose veins in Albania: a retrospective, single-centre analysis. Arch Med Sci Atheroscler Dis 2024; 9: e41-6. doi:10.5114/AMSAD/183653 https://doi.org/10.5114/amsad/183653 PMid:38846060 PMCid:PMC11155458 |
||||
| 15.Mok Y, Ballew SH, Kucharska-Newton A, Butler K, Henke P, Lutsey PL, et al. Demographic and clinical risk factors of developing clinically recognized varicose veins in older adults. Am J Prev Med 2025; 68: 674-81. doi:10.1016/J.AMEPRE.2024.12.009 https://doi.org/10.1016/j.amepre.2024.12.009 PMid:39701487 PMCid:PMC11925675 |
||||
| 16. Fusco I, Granata G, Trombetta S, Mancusi C, Genua M, Oddi FM. Primary care-based assessment of chronic venous insufficiency: a cross-sectional analytical study. Veins Lymphat 2026; 15: 14891. doi:10.4081/vl.2026.14891 https://doi.org/10.4081/vl.2026.14891 |
||||
| 17.Fadlallah H, El Masri D, Bahmad HF, Abou-Kheir W, El Masri J. Update on the Complications and management of liver cirrhosis. Med Sci (Basel) 2025; 13. doi:10.3390/MEDSCI13010013 https://doi.org/10.3390/medsci13010013 PMid:39982238 PMCid:PMC11843904 |
||||
| 18.Doğan K, Çetinkaya F, Taş A, Doğan K, Çetinkaya F, Taş A, et al. Link between lower extremity venous reflux and varicocele in adult male patients: A prospective study. Cardiovasc Surg Interv 2024; 11: 108-13. doi:10.5606/E-CVSI.2024.1666 https://doi.org/10.5606/e-cvsi.2024.1666 |
||||
| 19.Clark MR, Taylor AC. Pelvic venous disorders: An update in terminology, diagnosis, and treatment. Semin Intervent Radiol 2023; 40: 362-71. doi:10.1055/S-0043-1771041 https://doi.org/10.1055/s-0043-1771041 PMid:37575340 PMCid:PMC10415053 |
||||
| 20.Previtali E, Bucciarelli P, Passamonti SM, Martinelli I. Risk factors for venous and arterial thrombosis. Blood Transfus 2011; 9: 120-38. doi:10.2450/2010.0066-10 | ||||
| 21.Gavrilov S, Moskalenko YP, Mishakina NY, Efremova OI, Kulikov VM, Grishenkova AS. Stratification of pelvic venous reflux in patients with pelvic varicose veins. J Vasc Surg Venous Lymphat Disord 2021; 9: 1417-24. doi:10.1016/J.JVSV.2021.04.019 https://doi.org/10.1016/j.jvsv.2021.04.019 PMid:34023538 |
||||
| 22.Wang K, Siegel L, Betancourt A, Keefe NA, Salazar G, Browder S, et al. Frequency of left common iliac vein compression in asymptomatic adolescents and young adults. J Vasc Surg Venous Lymphat Disord 2025; 13: 102282. doi:10.1016/J.JVSV.2025.102282 https://doi.org/10.1016/j.jvsv.2025.102282 PMid:40545194 PMCid:PMC12311583 |
||||
| 23.Zhang W, Lan F, Yuan S, Guo J, Li Y, Pan Y, et al. Diagnostic and therapeutic impact of ultrasound tomography in lower extremity varicose veins: protocol for a paired accuracy study and randomised controlled trial in a tertiary hospital in China. BMJ Open 2025; 15. doi:10.1136/BMJOPEN-2025-107744 https://doi.org/10.1136/bmjopen-2025-107744 PMid:41033759 PMCid:PMC12496035 |
||||
| 24.Yang X, Zhang X, Yin M, Wang R, Lu X, Ye K. Elastic compression stockings to prevent post-thrombotic syndrome in proximal deep venous thrombosis patients without thrombus removal. J Vasc Surg Venous Lymphat Disord 2022; 10: 293-9. doi:10.1016/j.jvsv.2021.06.023 https://doi.org/10.1016/j.jvsv.2021.06.023 PMid:34358673 |
||||
| 25.Rossi FH, Rodrigues TO, Izukawa NM, Kambara AM. Best practices in diagnosis and treatment of chronic iliac vein obstruction. J Vasc Bras 2020; 19. doi:10.1590/1677-5449.190134 https://doi.org/10.1590/1677-5449.190134 PMid:34290748 PMCid:PMC8276648 |
||||
| 26.Heller T, Teichert C, Hafer J, Weber MA, Kröger JC, Meinel FG. Prevalence of May-Thurner syndrome in patients with deep vein thrombosis at a large medical referral center. Rofo 2019; 191: 1107-17. doi:10.1055/A-0959-6230 . https://doi.org/10.1055/a-0959-6230 PMid:31272108 |
||||
| 27.Osman AM, Mordi A, Khattab R. Female pelvic congestion syndrome: how can CT and MRI help in the management decision? Br J Radiol 2021; 94. doi:10.1259/BJR.20200881 https://doi.org/10.1259/bjr.20200881 PMid:33252986 PMCid:PMC7934308 |
||||
| 28.Corrêa MP, Kurtz GS, Bianchini L, Copatti L, Ribeiro M, Saleh JN, et al. Prevalence of left iliac vein compression on computed tomography scans from a population. J Vasc Bras 2020; 19: e20190060. doi:10.1590/1677-5449.190060 https://doi.org/10.1590/1677-5449.190060 PMid:34178055 PMCid:PMC8202169 |
||||
| 29.Badawy E, Seif MA, Elmoheen A. May-Thurner syndrome: A neglected cause of unilateral leg swelling. Open Access Emerg Med 2020; 12: 121-5. doi:10.2147/OAEM.S246018 https://doi.org/10.2147/OAEM.S246018 PMid:32431556 PMCid:PMC7202772 |
||||
| 30.Uppot RN, Sahani D V., Hahn PF, Gervais D, Mueller PR. Impact of obesity on medical imaging and image-guided intervention. AJR Am J Roentgenol 2007; 188: 433-40. doi:10.2214/AJR.06.0409 https://doi.org/10.2214/AJR.06.0409 PMid:17242253 |
||||
| 31.Gagne PJ, Gasparis A, Black S, Thorpe P, Passman M, Vedantham S, et al. Analysis of threshold stenosis by multiplanar venogram and intravascular ultrasound examination for predicting clinical improvement after iliofemoral vein stenting in the VIDIO trial. J Vasc Surg Venous Lymphat Disord 2018; 6: 48-56.e1. doi:10.1016/j.jvsv.2017.07.009 https://doi.org/10.1016/j.jvsv.2017.07.009 PMid:29033314 |
||||
| 32.Clode H, Spencer EB, Nelson J, Horne ES. Efficacy of ultrasound in the evaluation of iliac vein stenting. Phlebology 2025; 40: 110-5. doi:10.1177/02683555241276565 https://doi.org/10.1177/02683555241276565 PMid:39186827 |
||||
Copyright

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
AUTHOR'S CORNER

